DOP15 Transmural healing with vedolizumab in patients with active Crohn’s disease
J. Rimola1, J.F. Colombel2, B. Bressler3, S. Adsul4, J. Siegelman5, P.E. Cole6, D. Lindner7, S. Danese8
1Department of Radiology, Hospital Clínic de Barcelona, Barcelona, Spain, 2Icahn School of Medicine at Mount Sinai, Department of Gastroenterology, New York, USA, 3Division of Gastroenterology-, University of British Columbia, Vancouver, Canada, 4Takeda, Global Medical Affairs, Zurich, Switzerland, 5Clinical Development and Clinical Translation, Takeda, Cambridge, USA, 6Takeda, Department of Imaging, Cambridge, USA, 7Takeda, Statistical and Quantitative Sciences, Zurich, Switzerland, 8Department of Gastrointestinal Immunopathology, Humanitas University, Milan, Italy
Background
VERSIFY, an open-label phase 3b study of vedolizumab (NCT02425111; EudraCT 2014-003509-13), demonstrated endoscopic mucosal healing in patients with Crohn’s disease (CD).1 An exploratory analysis of a subset of these patients who underwent magnetic resonance enterography (MREn) also demonstrated healing.1 Further exploratory analysis of MREn imaging features of transmurality in patients treated with vedolizumab is reported herein.
Methods
In VERSIFY, patients treated with intravenous vedolizumab (300 mg) on Day 1, Weeks 2 and 6, and then every 8 weeks for 26 or 52 weeks were imaged at screening, Week 26, and Week 52, with images assessed centrally.1 All patients with disease detectable by MREn at screening (≥1 ileocolonic segment with a segmental magnetic resonance index of activity [MaRIA] score ≥7) who completed 26 or 52 weeks of treatment and had evaluable segments at all MREn timepoints were included in this analysis. Ileocolonic segments with a MaRIA score ≥7 at baseline were evaluated for improvement of oedema and bowel wall thickness. Further exploratory assessment of mesenteric fluid and lymph nodes are ongoing.
Results
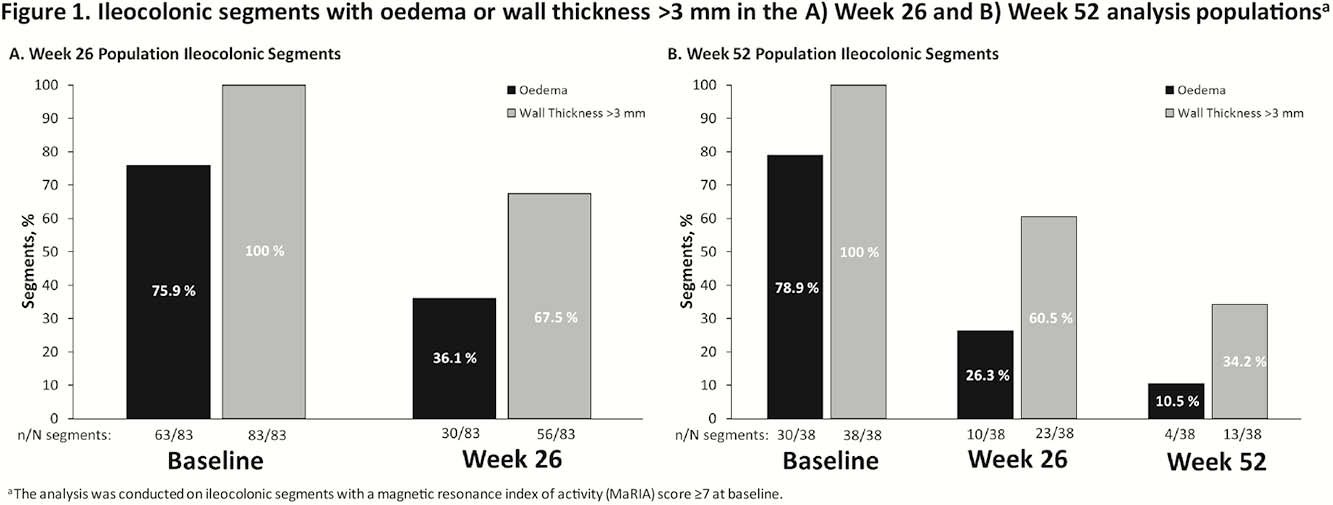
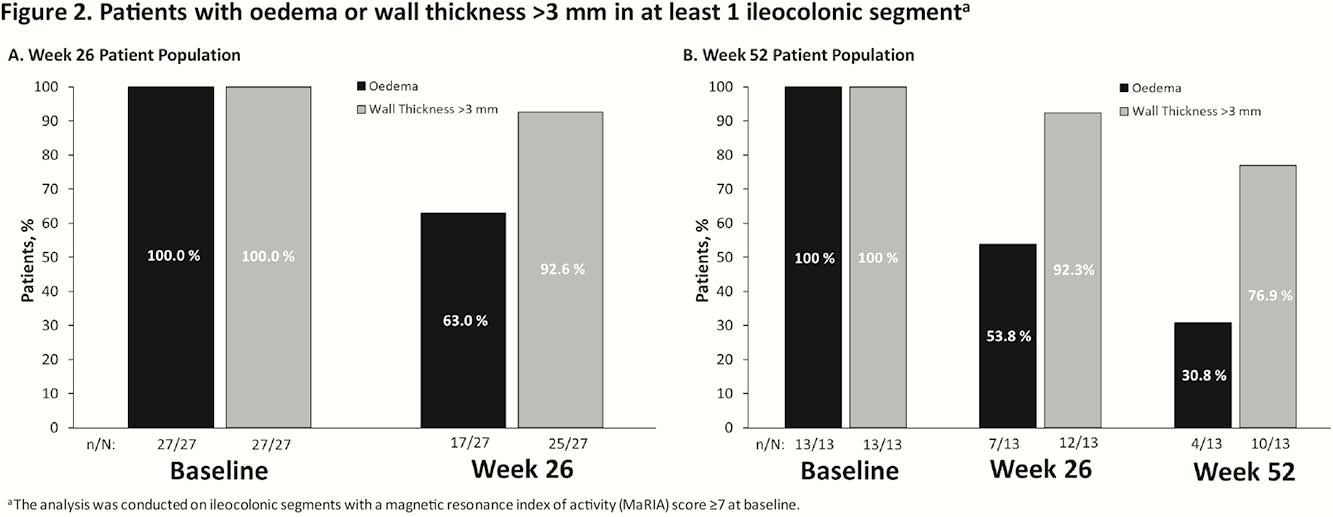
Week 26 analyses included 27 patients with a total of 83 ileocolonic segments; Week 52 analyses included 13 patients with a total of 38 segments. Week 26 and Week 52 patient populations had baseline mean (standard deviation [SD]) simplified endoscopic activity scores for CD (SES-CD) of 15.7 (7.7) and 16.5 (7.8) and Crohn’s disease activity index (CDAI) scores of 301 (61) and 297 (79), respectively. The proportion of segments with oedema or wall thickness >3 mm decreased from baseline to Week 26 and to Week 52 (Figure 1). All segments evaluated had wall thickness >3 mm at baseline. The proportion of patients with oedema or wall thickness >3 mm in ≥1 evaluated segment decreased from baseline to Week 26 and/or Week 52 (Figure 2). In the Week 26 population, 2/27 (7.4%) patients had no oedema with wall thickness <3 mm at Week 26 in segments that had MaRIA ≥7 at baseline. In the Week 52 population, 1/13 (7.7%) patients at Week 26 and 3/13 (23.1%) at Week 52 had no oedema with wall thickness <3 mm in segments that had MaRIA ≥7 at baseline.
Conclusion
Vedolizumab treatment of patients with CD reduced the number of ileocolonic segments with transmural inflammation (oedema, wall thickness >3 mm) as detected by MREn and the overall number of patients with transmural inflammation from baseline at treatment Weeks 26 and 52. This exploratory analysis contributes to the understanding of transmural disease processes in CD and the impact of vedolizumab on components of transmural inflammation in CD.
Danese S,


