DOP19 Real-world healthcare resource utilisation among patients with inflammatory bowel disease administered vedolizumab for 6 months
B. Cohen1, H. Chang2, J.R. Jason Rogers1, S. Garg1, D. Lawrence3, F. Fasihuddin1, T. Lissoos4, A. Atreja5
1Department of Gastroenterology, Icahn School of Medicine at Mount Sinai, New York, USA, 2Department of Population Health Science and Policy, Icahn School of Medicine at Mount Sinai, New York, USA, 3Department of Gastroenterology, Takeda Pharmaceuticals, Deerfield, USA, 4Lilly Biomedicines, Eli Lilly & Company, Indianapolis, USA, 5Department of Gastroenterology, Mount Sinai, New York, USA
Background
Inflammatory bowel disease (IBD) is associated with high healthcare resource utilisation. Vedolizumab (VDZ) is a gut-selective monoclonal antibody that binds the α4β7 integrin and is indicated for the treatment of adult patients with moderately to severely active ulcerative colitis (UC) or Crohn’s disease (CD). We investigated real-world healthcare resource utilisation for these patients in the 6 months after initiating VDZ treatment.
Methods
This retrospective, non-interventional, cohort study examined healthcare resource utilisation for adults with IBD treated with VDZ at Mt Sinai Medical Center (New York, NY) between June 1, 2014, and May 31, 2019. IBD-related healthcare resource utilisation (emergency room [ER] visits, hospitalisations, procedures, corticosteroids) and VDZ treatment patterns (induction, persistence, adherence) were assessed for 6 months after first VDZ infusion. Discontinuation was defined as a ≥90-day gap between infusions or stopping VDZ treatment <6 months after the first infusion. Patients who had ≥90-day gaps were not counted as discontinuing if they experienced clinical benefit that continued beyond the 6-month period.
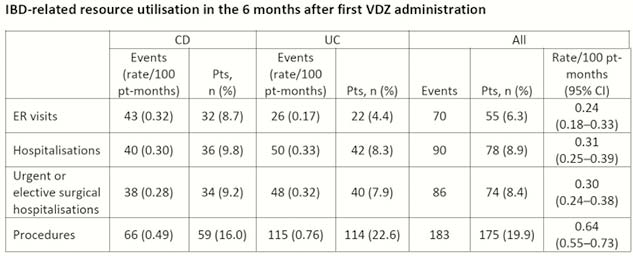
Results
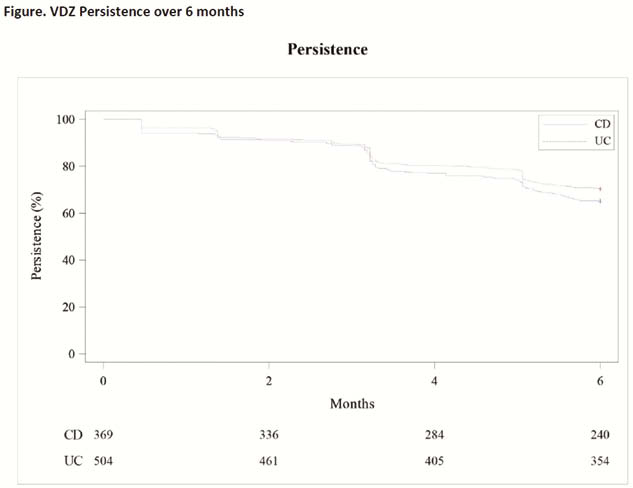
880 patients treated with VDZ were assessed. Mean age was 39.6 years; 51.6% of patients were female. 369 (41.9%) and 504 (57.3%) patients were diagnosed with CD and UC, respectively. 81.4% of patients received 3 VDZ induction doses by Day 98; 69.0% received 4 doses by Day 120. The rates of ER visits, hospitalisations and elective surgical or urgent hospitalisations and procedures among VDZ-treated patients were <1 per 100 pt–months. Steroids (oral or IV) were prescribed to 42.2% of patients during the 6-month period. In the 2 months after their last infusion, 81.6% of patients who received ≥3 VDZ doses were steroid-free. Timing of the last infusion was variable. Overall VDZ persistence was 68.0% (65.0% and 70.2% for CD and UC, respectively; Figure). Most patients (67.2%) had ≥80% of days covered in a 180-day period
Conclusion
At a tertiary IBD centre, VDZ was associated with low healthcare resource utilisation during the first 6 months of treatment. More than two-thirds of patients persisted with treatment, with most being steroid-free at last follow-up.


