DOP41 Low-grade dysplasia prognosis and predictive factors for advanced neoplasia progression in the 21st century: A large multi-centre retrospective cohort study
M. Kabir1,2, K. Curtius3, I. Al-Bakir2,3,4, J. Hartono5, M. Johnson6, J.E. East6, J.O. Lindsay7,8, R. Vega9, S. Thomas-Gibson1,2, A. Hart2,4, T. Graham3, A. Wilson1,2
1Wolfson Endoscopy Unit, St Mark’s Hospital, London, UK, 2Division of Surgery and Cancer, Imperial College London, London, UK, 3Barts Cancer Institute, Queen Mary University of London, London, UK, 4Inflammatory Bowel Diseases Unit, St Mark’s Hospital, London, UK, 5Gastroenterology Department, National University Hospital, Singapore, 6Translational Gastroenterology Unit, Nuffield Department of Medicine, John Radcliffe Hospital, University of Oxford, Oxford, UK, 7Department of Gastroenterology, Royal London Hospital, Barts Health NHS Trust, London, UK, 8Barts and the London School of Medicine and Dentistry, Centre for Immunobiology, Queen Mary University of London, London, UK, 9Endoscopy Unit, University College London Hospital, London, UK
Background
Recent advances in ulcerative colitis (UC) endoscopic surveillance such as high-definition imaging and greater chromoendoscopy (CE) use have led to an increase in detection and resection of visible dysplasia. An updated study of prognosis of low-grade dysplasia (LGD) is needed to address uncertainty as to the accuracy of progression rates based on historical studies.
Methods
This retrospective cohort study involved four UK IBD centres. Hospital and endoscopy pathology databases were searched between 1 January 2001 and 30 December 2018 to identify adult patients with UC who had their first LGD diagnosis diagnosed within the extent of colitis. Only patients followed up with at least one colonoscopy or colectomy by 30 August 2019 were included. The study endpoint was time to high-grade dysplasia (HGD) or cancer (CRC), i.e. advanced neoplasia (AN), or end of follow-up. Survival analyses were performed using Kaplan–Meier estimation and Cox proportional hazards (PH) models.
Results
In total, 460 patients met the inclusion criteria and were followed up for a median of 4.1 years (IQR 6), equating to 2,232 patient-years. A mean of 3.7 (range 0–17) subsequent colonoscopies was performed per patient. Seventy-seven per cent of patients had CE surveillance. Complete endoscopic resection was achieved in 94% and 64% of the polypoid and non-polypoid LGD, respectively. There was progression to AN in 88 cases (19%) during follow-up. There was no significant difference in AN progression between centres. Unresectable non-polypoid or invisible LGD carried the greatest risk of AN development (Figure 1).
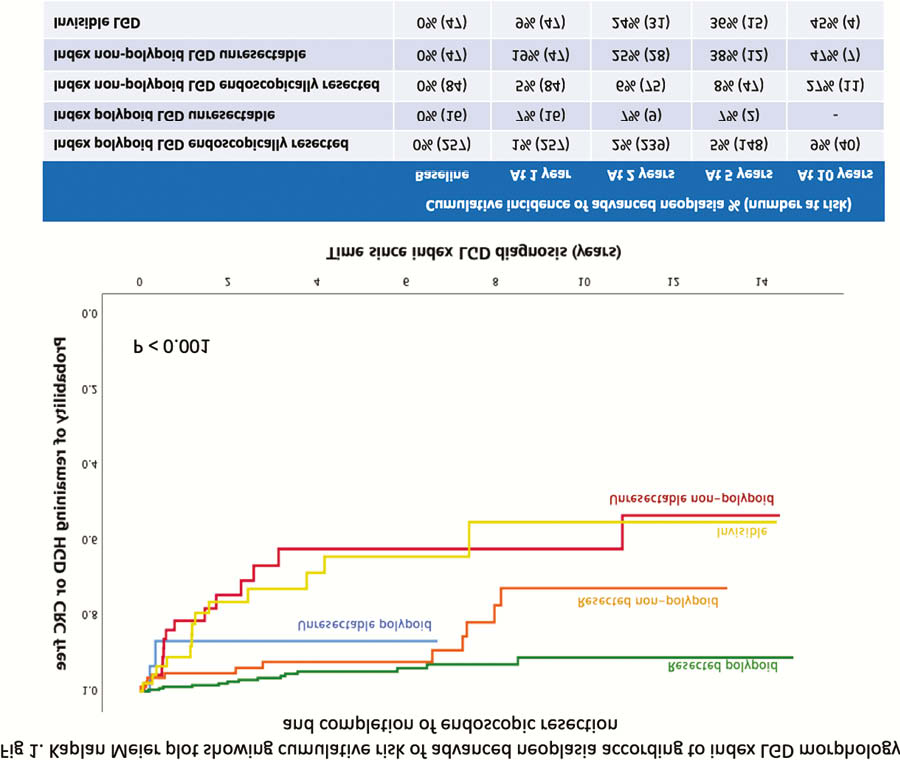
On univariate Cox PH analysis, CE use was protective against AN progression (HR 0.5; 95% CI 0.3–1.0;
| Variable | Hazard ratio (95% CI) | |
| Index LGD characteristics | ||
| Visible, size < 1cm | 1 | <0.001 |
| Visible, size 1cm or more | 3.8 (1.8–7.9) | 0.048 |
| Invisible | 2.8 (1.0–7.5) | |
| Complete endoscopic resection of index LGD | 0.3 (0.1–0.5) | <0.001 |
| Multifocal LGD | 2.1 (1.1–3.8) | 0.016 |
| Moderate/severe histological inflammation within last 5 years up to LGD diagnosis | 1.4 (0.8–2.5) | 0.229 |
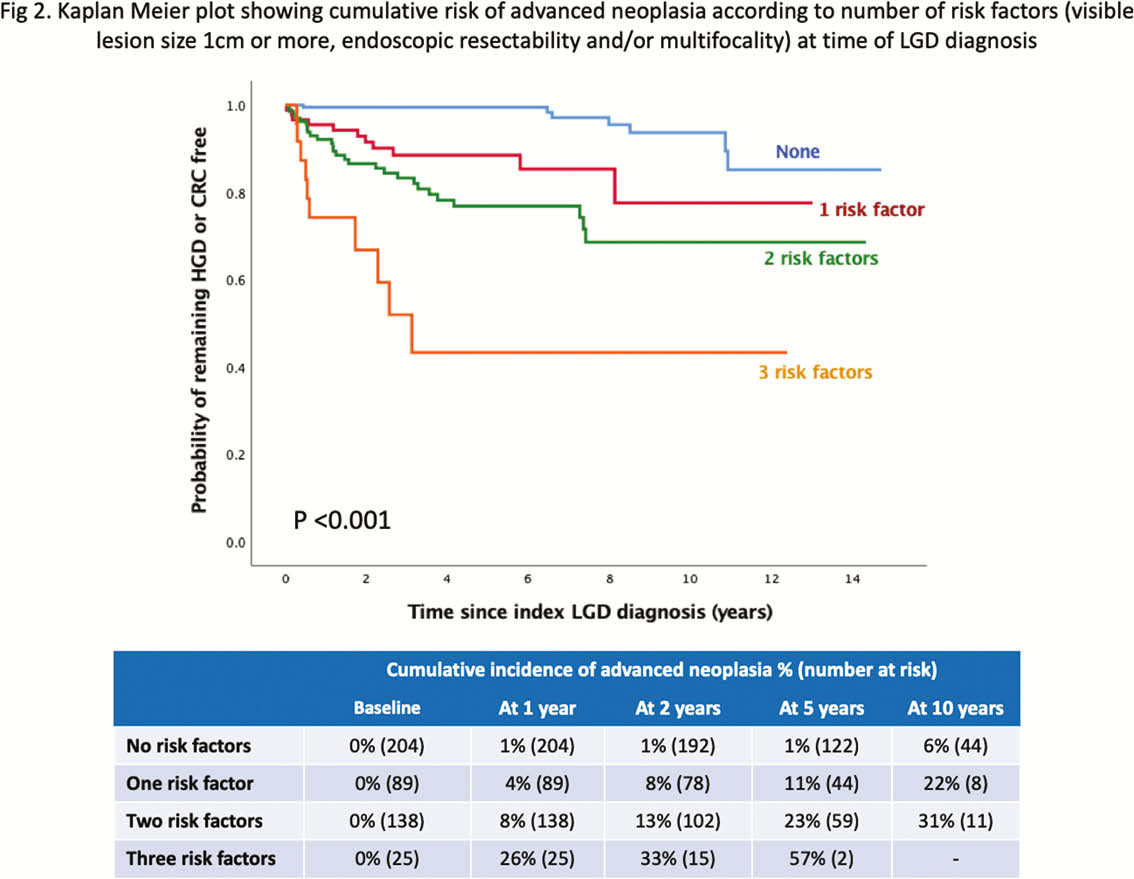
Cumulative risk of AN increased with the number of risk factors (Figure 2).
Conclusion
This is the largest study examining prognosis of LGD, based on endoscopic features, in this century. Five-year cumulative incidence of AN is low after complete endoscopic resection of visible LGD without surrounding dysplasia. Lesion size of 1 cm or more, invisibility, multifocality and unresectability of LGD are significant risk factors for progression to AN. These factors should be taken into consideration when discussing management options with patients.


