DOP57 Vedolizumab has similar efficacy rates in young and elderly patients: A binational, multicentre, retrospective case–control study
N.A. Cohen1,2, N. Plevris3, U. Kopylov2,4, A. Grinman2,4, B. Ungar1,2, H. Yanai2,5, H. Leibovitzh2,5, N. Fliss1,2, A. Hirsch1,2, E. Ritter1,2, Y. Ron1,2, A. Bar-Gil Shitrit6,7, E. Goldin6,7, I. Dotan2,5, S. Ben-Horin2,4, C. Lees3,8, N. Maharshak1,2
1Department of Gastroenterology and Liver Diseases, Tel Aviv Medical Center, Tel Aviv, Israel, 2Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel, 3The Edinburgh IBD Unit, Western General Hospital, Edinburgh, UK, 4Department of Gastroenterology and Liver Diseases, Sheba Medical Center, Ramat Gan, Israel, 5IBD Center, Division of Gastroenterology, Rabin Medical Center, Petah Tikvah, Israel, 6Digestive Diseases Institute, Shaare Zedek Medical Center, Jerusalem, Israel, 7Hadassah Medical School, Hebrew University of Jerusalem, Jerusalem, Israel, 8The University of Edinburgh, Western General Hospital Campus, Center for Genomic and Experimental Medicine, MRC Institute of Genetics and Molecular Medicine, Edinburgh, UK
Background
Treatment of inflammatory bowel diseases (IBD) is associated with multiple side effects, such as malignancies and infections, that may be more common in the elderly population. The aim of this study was to compare the safety and efficacy of vedolizumab (VDZ) in young and elderly patients.
Methods
This is a binational, multicentre, retrospective, case–control study consisting of patients from multiple centres in Israel and from Edinburgh, UK. IBD patients in whom VDZ treatment was initiated during 2015–2019, and were followed for at least 14 weeks, were included in the study according to their age groups: ≤40 years or ≥60 years. Patients were matched for disease duration, classification and severity.
Results
We recruited 144 patients (82 CD and 62 UC) to the elderly cohort and 140 patients (83 CD and 57 UC) to the young cohort. The respective average age was 70.2 ± 7.3 years and 29.6 ± 5.7 years. Elderly patients had significantly more cardiovascular, metabolic and respiratory co-morbidities and had less prior exposure to anti-TNF agents compared with the young cohort (CD: 51% vs. 88%,
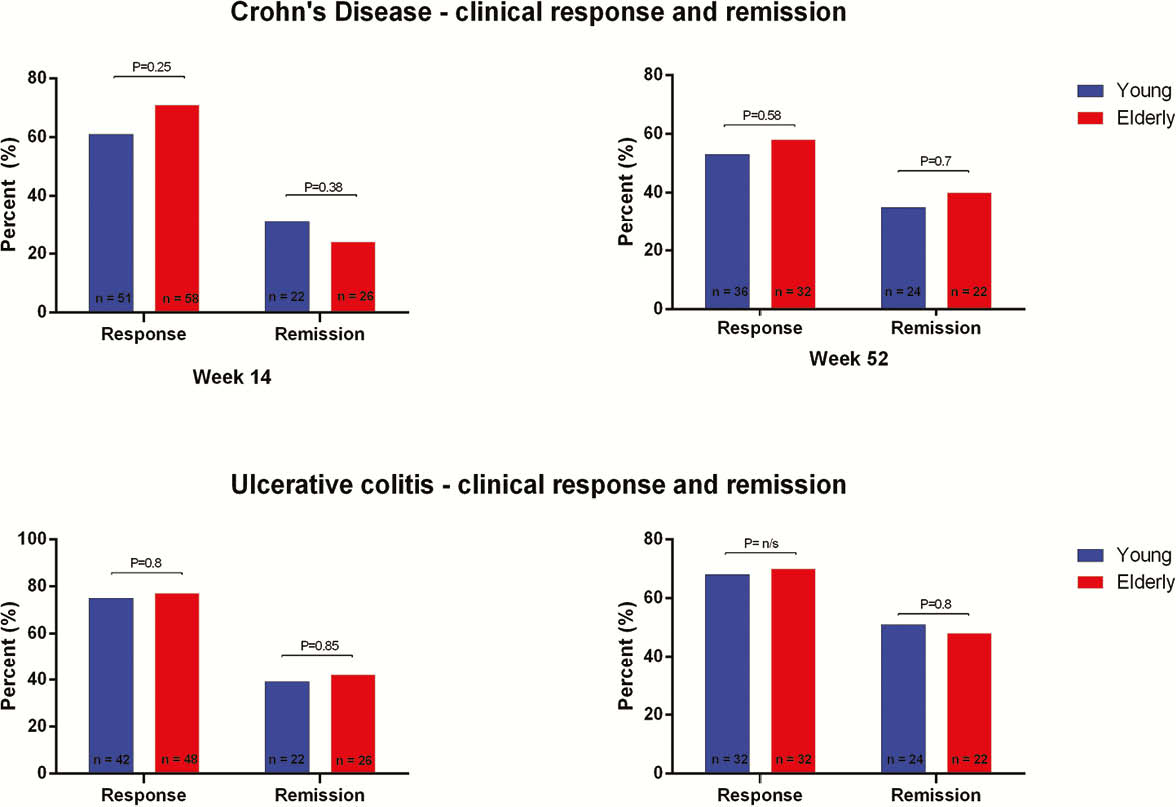
Conclusion
VDZ is equally effective in elderly and young IBD patients. This study shows an increased risk of overall infections in the elderly cohort, it is unclear whether these are related to VDZ treatment or the age and background diseases of these patients. In elderly patients, the use of VDZ before anti-TNF therapy may improve response and remission rates.


