DOP60 Vedolizumab treatment persistence and safety in an extended access program (XAP)
S. Danese1, K. Subramanian2, J. Van Zyl3, S. Adsul4, D. Lindner5, J. Roth6, S. Vermeire7
1Department of Gastrointestinal Immunopathology, Humanitas University, Milan, Italy, 2Gastroenterology and Hepatology Unit, Canberra Hospital, Canberra, Australia, 3University of the Free State, Department of Gastroenterology, Bloemfontein- Free State, South Africa, 4Department of Global Medical Affairs, Takeda Pharmaceuticals International AG, Zurich, Switzerland, 5Statistical and Quantitative Sciences, Data Sciences Institute, Takeda Pharmaceuticals International AG, Zurich, Switzerland, 6Chief Medical Office, Global Medical Research, Takeda Pharmaceuticals International AG, Zurich, Switzerland, 7Department of Gastroenterology, University Hospitals Leuven, Leuven, Belgium
Background
Vedolizumab, a gut-selective, α 4β 7 integrin antagonist, has been established as an effective and safe treatment for patients with UC or CD in the GEMINI phase 3 program and long-term safety (LTS) study. An extended access program (XAP) was initiated to provide continued access to patients who were benefiting from vedolizumab in GEMINI LTS and to monitor safety. We now report persistence and safety results from a 2-year data analysis.
Methods
Vedolizumab XAP (NCT02743806) is a phase 3b/4 prospective, open-label, multinational interventional study. A rollover from GEMINI LTS (NCT00790933) to the XAP, patients reduced dosing frequency from vedolizumab 300 mg IV every 4 weeks (Q4W) to every 8 weeks (Q8W) or remained on vedolizumab 300 mg IV Q4W if medically indicated. This 2-year data analysis assessed persistence on Q8W dosing after dosing frequency reduction, need for escalation to Q4W dosing, incidence of relapse (defined as dose escalation, study withdrawal due to adverse event, loss of adequate benefit from vedolizumab, or increased corticosteroid [CS] or immunomodulator dose), and safety 2 years after rollover from GEMINI LTS.
Results
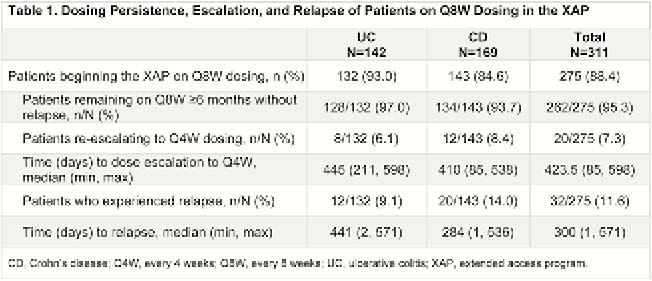
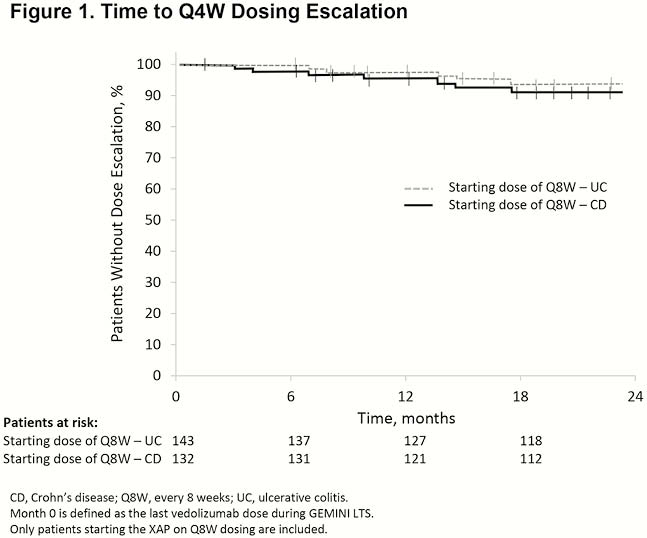
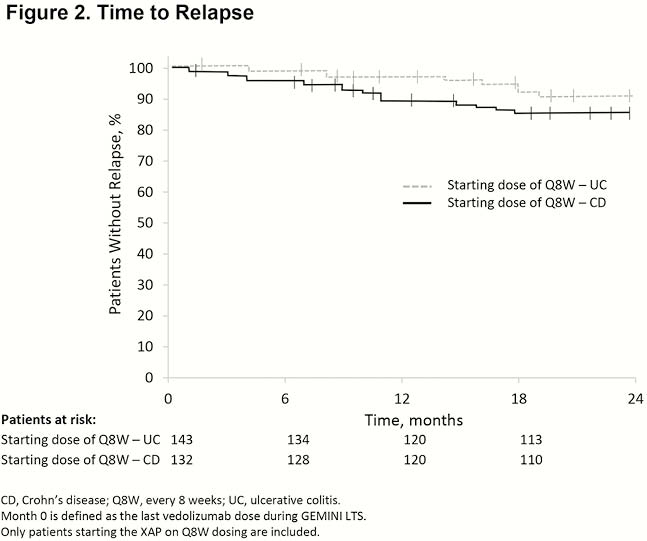
A total of 311 patients (142 UC, 169 CD) from GEMINI LTS enrolled in the XAP. Median (range) duration of exposure to vedolizumab prior to the XAP was 8.0 years (5.2–10.0) for patients with UC and 7.5 years (5.4–9.9) for patients with CD. Of patients with UC and CD, 93.0% and 84.6%, respectively, reduced dosing frequency to Q8W at XAP start, and 87.3% and 77.5%, respectively, remained on Q8W after 2 years. At baseline, 93.0% of all patients with UC and 88.2% of all patients with CD were in CS-free remission; patients who maintained Q4W dosing at baseline had lower CS-free remission rates (62.5% in UC and 69.2% in CD). Of patients who initiated Q8W dosing at enrolment in the XAP, 95% had no relapse for ≥6 months (97.0% UC, 93.7% CD; Table 1). Only 7.3% of patients required dose escalation to Q4W, and 11.6% experienced relapse during the 2-year follow-up. Times to dose escalation and relapse were similar in patients with UC and CD (Figures 1 and 2). At 2 years, 4 of 8 patients with UC and 4 of 12 patients with CD who required dose escalation to Q4W discontinued vedolizumab early due to loss of benefit or adverse events. Adverse events related to vedolizumab were infrequent; no new or serious events attributed to vedolizumab were reported.
Conclusion
High patient persistence on Q8W vedolizumab was observed in the first 2 years after reduction of dosing frequency in the XAP. Overall, there were low rates of Q4W dose escalation and CD or UC disease relapse. The safety profile was consistent with previous reports with no new signals observed.


