DOP61 Tofacitinib, an oral, small-molecule Janus kinase inhibitor, in the treatment of ulcerative colitis: Analysis of an open-label, long-term extension study with up to 5.9 years of treatment
G.R. Lichtenstein1, E.V. Loftus Jr2, S.C. Wei3, A.O. Damião4, D. Judd5, N. Lawendy5, G. Chan5, H. Zhang5, W. Wang5, A.J. Thorpe5, S. Bloom6
1Division of Gastroenterology, Perelman School of Medicine at University of Pennsylvania, Philadelphia, USA, 2Division of Gastroenterology and Hepatology, Mayo Clinic, Rochester, MN, USA, 3Department of Internal Medicine, National Taiwan University Hospital and College of Medicine, Taipei, Taiwan, Province of China, 4Department of Gastroenterology, University of São Paulo School of Medicine, São Paulo, Brazil, 5Pfizer Inc., Collegeville, PA, USA, 6University College London Hospitals NHS Foundation Trust, London, UK, UK
Background
Tofacitinib is an oral, small-molecule JAK inhibitor for the treatment of ulcerative colitis (UC). Efficacy and safety of tofacitinib were demonstrated in patients with moderate to severe UC in 3 Phase 3 studies.1 Here, we present data from an ongoing, open-label, long-term extension (OLE) study.2
Methods
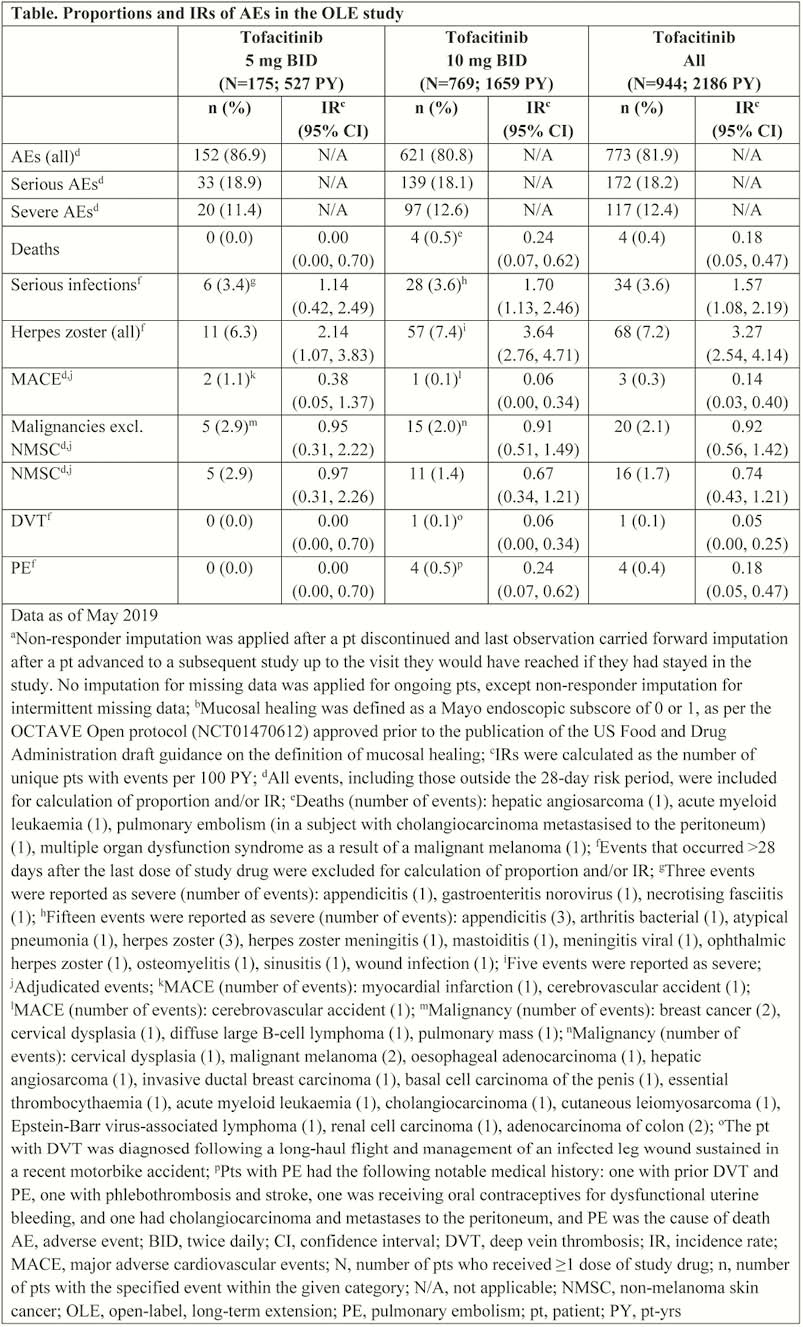
We present updated safety and efficacy data from the OLE study (OCTAVE Open, NCT01470612; as of May 2019, database not locked). Eligible patients included non-responders (Week 8 data) in OCTAVE Induction 1 and 2 (NCT01465763; NCT01458951) and completers (Week 52 data) or treatment failures (early-termination data) in OCTAVE Sustain (NCT01458574). Patients in remission (total Mayo score ≤2, no individual subscore >1, rectal bleeding [RB] subscore 0) at Week 52 of OCTAVE Sustain (central read) received tofacitinib 5 mg twice daily (BID); all others received 10 mg BID. Induction non-responders without clinical response (≥3-point and ≥30% decrease from induction study baseline total Mayo score, plus ≥1-point RB subscore decrease or absolute RB subscore ≤1) at Month 2 of the OLE study were withdrawn. Incidence rates (IRs) for adverse events (AEs) of special interest were calculated (no. of unique patients with events per 100 patient-years). Efficacy endpoints were derived from Mayo score (local read) with non-responder and last observation carried forward imputation (NRI-LOCF) [a].
Results
Of 944 patients who received ≥1 dose of tofacitinib, 769 (81.5%) received 10 mg BID (median duration [range]: 5 mg BID 1170 [36–2066]; 10 mg BID 668 [1–2159] days). In total, 338 (35.8%) and 93 (9.9%) patients discontinued due to insufficient clinical response and AEs (excl. worsening UC), respectively. IRs (95% confidence interval) in the Tofacitinib. All group were: deaths 0.18 (0.05, 0.47); serious infections 1.57 (1.08, 2.19); herpes zoster (non-serious and serious) 3.27 (2.54, 4.14); major adverse cardiovascular events 0.14 (0.03, 0.40); malignancies excl. non-melanoma skin cancer (NMSC) 0.92 (0.56, 1.42); NMSC 0.74 (0.43, 1.21); deep vein thrombosis 0.05 (0.00, 0.25); pulmonary embolism 0.18 (0.05, 0.47) (Table). At Month 36 (NRI-LOCF), 58.9% (
Conclusion
Incidence of AEs remained generally consistent in patients with moderate to severe UC in the OLE study compared with a previous analysis.2 Data continue to support long-term efficacy with tofacitinib up to 36 months beyond Week 52 of OCTAVE Sustain.
Sandborn WJ
Lichtenstein GR


