DOP71 Efficacy, safety, and tolerability of ustekinumab in paediatric patients with moderately to severely active Crohn’s disease: Results from, UniStar, a phase 1 study
J. Rosh1, D. Turner2, A. Griffiths3, D. Jacobstein4, O. Adedokun5, L. Padgett5, N. Terry5, C. O’Brien5, J. Hyams6
1Department of Pediatrics, Goryeb Children’s Hospital/Atlantic Health, Morristown, NJ, USA, 2Department of Pediatrics, Shaare Zedek Medical Center, Jerusalem, Israel, 3SickKids Hospital, Toronto, ON, Canada, 4Former Employee of Janssen Research and Development, LLC, Current Employee of Provention Bio, Research and Development, Horsham, PA, USA, 5Janssen Research and Development, LLC, Research and Development, Horsham, PA, USA, 6Department of Pediatrics, Connecticut Children’s Medical Center, Hartford, CT, USA
Background
Ustekinumab (UST) is approved for the treatment of adults with moderate to severe Crohn’s disease (CD). The objective of this study was to evaluate the pharmacokinetics (PK), safety, and efficacy of UST in paediatric patients with moderately to severely active CD who had failed treatment with corticosteroids (CS) and/or immunomodulators (IM) and/or anti-tumour necrosis factor (TNF) therapies. Here, we report the safety and efficacy results through Week 16; PK results are reported separately.
Methods
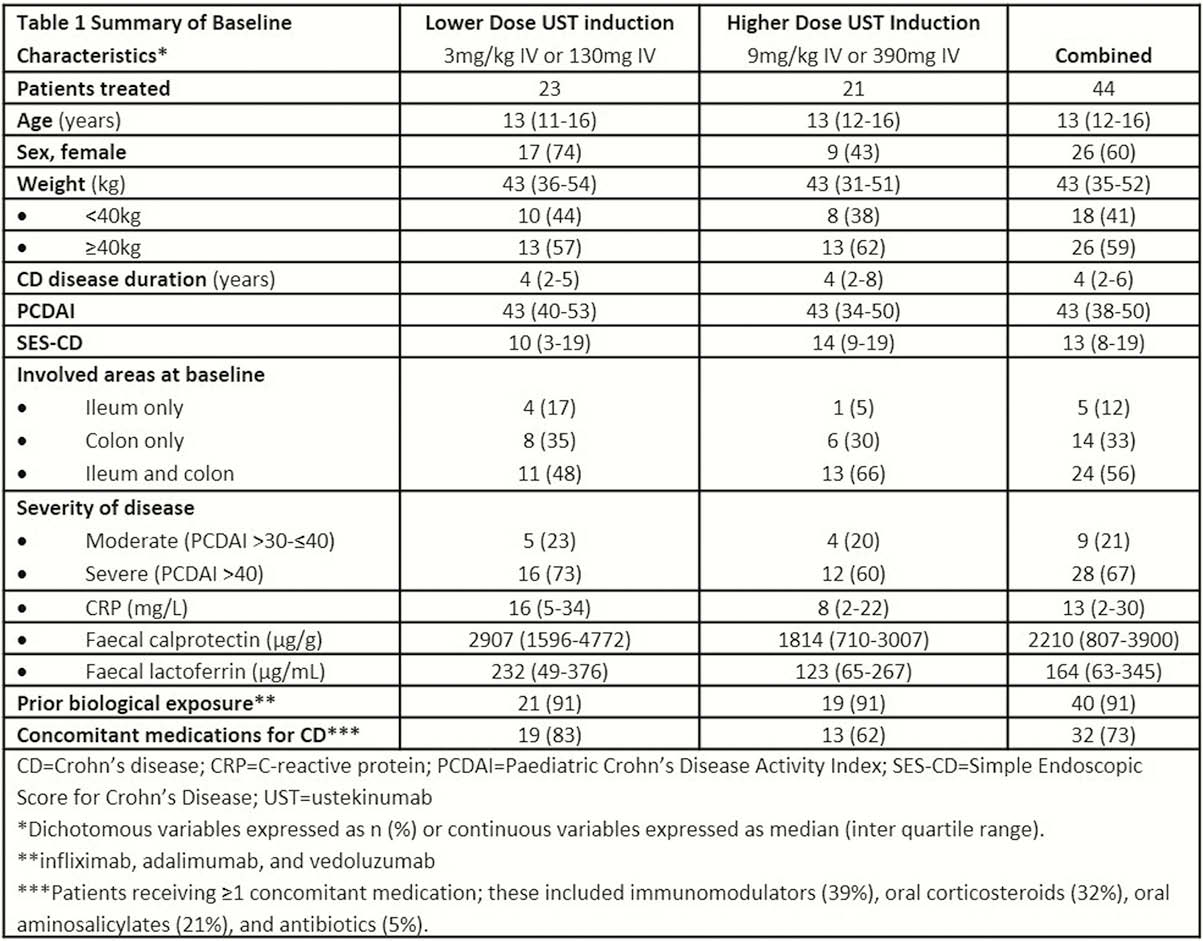
This was a Phase 1, multicentre, 16-week, double-blind induction dose-ranging study (NCT02968108) in patients aged 2 to <18 years (body weight [BW] ≥10 kg) with a Paediatric CD Activity Index (PCDAI) score >30 and at least an abnormal C-reactive protein >3.0 mg/l or faecal calprotectin >250 µg/g), or ulcerations in the ileum or colon upon ileocolonoscopy despite adequate treatment with CS ± IM ± anti-TNF therapy. Patients were randomised (1:1) and stratified by weight and prior anti-TNF use for induction to one of 2 weight range-based intravenous (IV) doses: 130mg vs. 390 mg if BW ≥40 kg and 3 mg/kg vs. 9 mg/kg if BW <40 kg. At week 8, all patients received a single subcutaneous (SC) UST maintenance dose of 90mg if BW ≥40 kg or 2 mg/kg if BW <40 kg.
Results
Forty-four patients (59% ≥40kg BW; >90% anti-TNF exposed) were randomised (
Conclusion
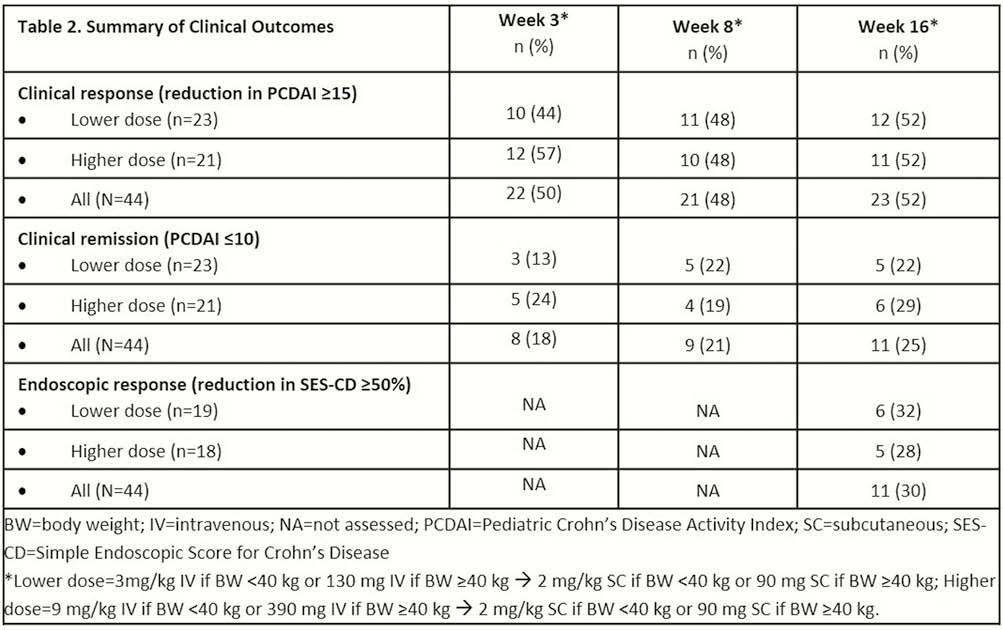
As early as 3 weeks and through 16 weeks, both the lower and higher doses of UST (IV week 0 and SC at week 8) improved clinical and endoscopic disease activity in this previously treatment-refractory group of children with CD. The safety profile was consistent with that for UST in adults with CD.


