DOP72 Safety, tolerability and efficacy of anti-TL1A antibody PF-06480605 in treatment of ulcerative colitis: The open-label, multicentre, Phase 2a TUSCANY study
S. Danese1, M. Klopocka2, E.J. Scherl3, J. Romatowski4, J.R. Allegretti5, E. Peeva6, M.S. Vincent6, U. Schoenbeck7, L. Xi6, Z. Ye6, M. Hassan-Zahraee6, N. Rath8, G. Li8, S. Neelakantan6, C. Banfield6, C. Lepsy9, D. Chandra6, K.E. Hung6
1Department of Gastroenterology, IBD Center, Humanitas Research Hospital, Milan, Italy, 2Department of Gastroenterology and Nutrition, Nicolaus Copernicus University in Toruń, Collegium Medicum, Bydgoszcz, Poland, 3Division of Gastroenterology and Hepatology, Jill Roberts Center for IBD, Weill Cornell Medicine, New York, NY, USA, 4Internal Medicine and Gastroenterology Department, J. Sniadecki’s Regional Hospital, Białystok, Poland, 5Division of Gastroenterology, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, USA, 6Pfizer Inc., Cambridge, MA, USA, 7Pfizer Inc., New York, NY, USA, 8Pfizer Inc., Collegeville, PA, USA, 9Pfizer Inc., Andover, MA, USA
Background
Widespread disruption of the mucosal immune system is central to the pathogenesis of ulcerative colitis (UC). One component, tumour necrosis factor (TNF) α-like ligand 1A (TL1A), is upregulated at the site of active disease in UC. Treatment of preclinical rodent models with anti-TL1A antibodies decreases disease activity, highlighting their therapeutic potential for UC. PF-06480605 is a first-in-class fully human immunoglobulin G1 monoclonal antibody targeting TL1A.
Methods
The Phase 2a, open-label, multicentre, single-arm TUSCANY study (NCT02840721) evaluated the safety, tolerability and efficacy of PF-06480605 in treatment of moderate to severe UC. Participants received 500 mg intravenous (IV) PF-06480605 every 2 weeks (Q2W) for a total of 7 doses, with a 14-week follow-up period. Primary safety and efficacy endpoints were incidence of treatment-emergent adverse events (TEAEs) and endoscopic improvement (EI) (Mayo endoscopic subscore [centrally read] of ≤1 without friability) at Week 14, respectively. Secondary efficacy endpoints included remission (total Mayo score ≤2, with no individual subscore >1) and endoscopic remission (Mayo endoscopic subscore = 0) at Week 14. Transcriptomic profiling on intestinal biopsies was performed.
Results
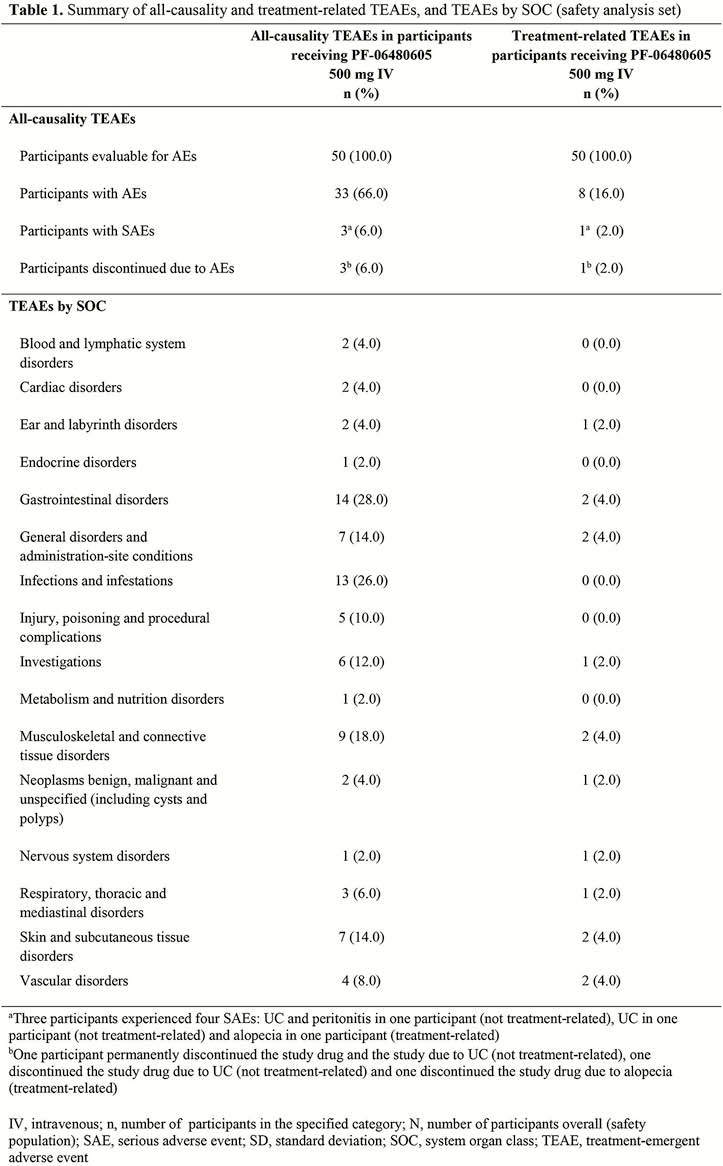
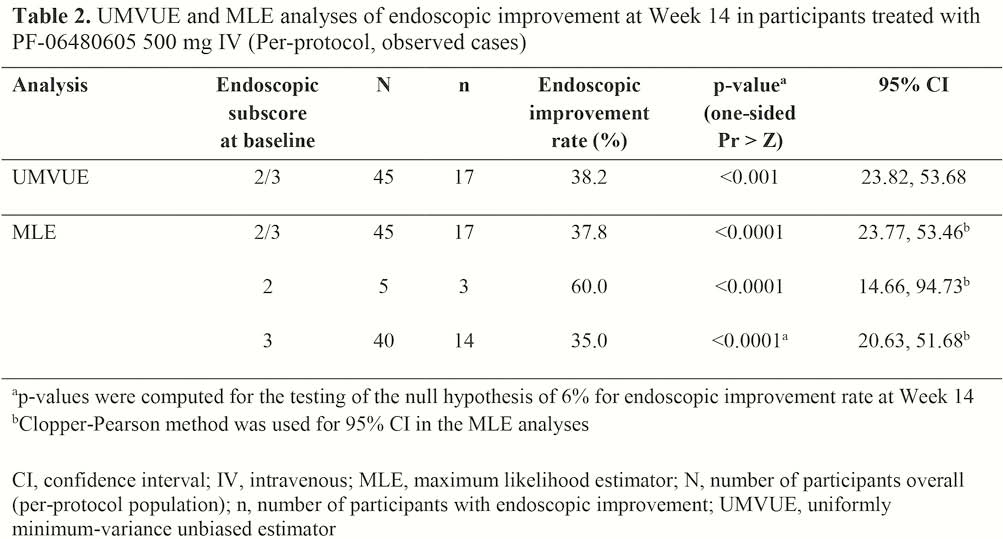
Of the 50 participants who received PF-06480605, 42 completed the study. The majority were male (56.0%) and white (96.0%), with a mean age of 40.0 years and prior experience of anti-TNF inhibitors (72.0%). Pancolitis (48.0%) and left-sided colitis (32.0%) were the most common forms of UC at baseline. There were 109 TEAEs, of which 18 were treatment-related. Aside from worsening UC, the most common TEAE by system organ class was arthralgia, which occurred in 6 participants, and 1 was treatment-related. Treatment-emergent serious adverse events were reported in 3 participants, and considered treatment-related in 1 participant (Table 1). No malignancies or deaths were reported. At Week 14, statistically significant EI was observed in 38.2% of participants (Table 2). The proportions of participants achieving remission and endoscopic remission at Week 14 were 24.0% and 10.0%, respectively. Transcriptomic analyses demonstrated normalisation towards a non-inflamed transcriptome in participants with EI.
Conclusion
PF-06480605 exhibited an acceptable safety and tolerability profile and statistically significant EI in participants with moderate to severe UC. These results warrant further evaluation in subsequent studies.


