DOP75 Efficacy of tofacitinib dose escalation to 10 mg BID in patients with UC after completing maintenance therapy in remission and losing response: Data from OCTAVE open-label, long-term extension study
D.T. Rubin1, M.C. Dubinsky2, J. Panés3, D.C. Wu4, N. Lawendy5, X. Guo5, C. Lai6, K. Kwok7, C. Su5, L. Salese5
1Inflammatory Bowel Disease Center, University of Chicago Medicine, Chicago, IL, USA, 2Icahn School of Medicine at Mount Sinai, New York, NY, USA, 3Hospital Clínic de Barcelona, IDIBAPS, CIBERehd, Barcelona, Spain, 4Division of Gastroenterology, Department of Internal Medicine, Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung, Taipei, Taiwan, Province of China, 5Pfizer Inc., Collegeville, PA, USA, 6Pfizer Inc., New Taipei City, Taiwan, Taiwan, Province of China, 7Pfizer Inc., New York, NY, USA
Background
Tofacitinib is an oral, small-molecule JAK inhibitor for the treatment of ulcerative colitis (UC). The efficacy and safety of tofacitinib were demonstrated in patients with moderate to severe UC in three Phase 3 studies.1 Here, we investigated outcomes in a subpopulation of the ongoing, open-label, long-term extension study (OLE) (OCTAVE Open; NCT01470612; May 2019; database not locked), including maintenance of remission patients receiving tofacitinib 5 mg twice daily (BID) who were dose-escalated to 10 mg BID due to flare.
Methods
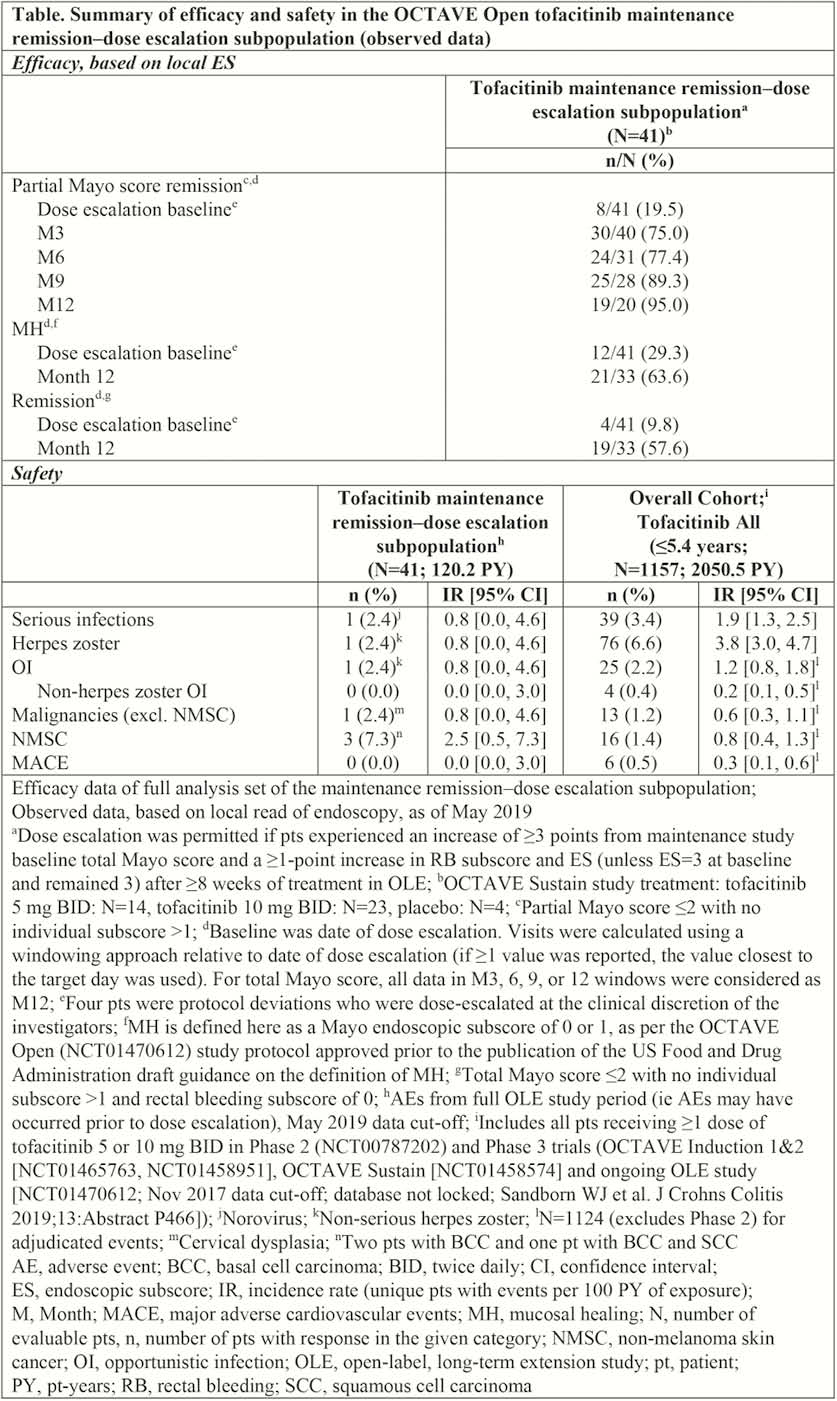
Analysis included the maintenance remission–dose-escalation subpopulation: patients who achieved remission at Week 52 in OCTAVE Sustain (tofacitinib 5 or 10 mg BID, or placebo), entered OLE (5 mg BID), but were dose-escalated to 10 mg BID due to flare (based on total Mayo score [MS]; local read endoscopy). Remission: total MS ≤2 with no subscore >1, and rectal bleeding (RB) subscore 0. Dose escalation during OLE was permitted if patients experienced a flare, defined as ≥3 increase from maintenance study baseline total MS and ≥1 increase in RB and endoscopic subscore (ES; unless ES=3 at baseline and remained 3) after ≥8 weeks of treatment in OLE. Kaplan–Meier for estimation of time to dose escalation was assessed in all OLE maintenance remission population relative to OLE baseline. Partial MS remission (≤2 with no subscore >1; no ES) assessed at Months (M)3, 6, 9 and 12 post-dose-escalation; mucosal healing (MH; ES ≤1) and remission assessed at M12 post-dose-escalation. Adverse events (AEs) also assessed.
Results
Of the 944 patients enrolled in OLE, 162 were in remission at baseline and received tofacitinib 5 mg BID, and 41 were dose-escalated to 10 mg BID due to flare (22 had prior TNFi failure). The cumulative proportion of patients with dose escalation was 4.3%, 10.6%, 12.6% and 13.2% at M3, 6, 9 and 12 of OLE, respectively. Partial MS remission was 75.0% at M3, and partial MS remission, MH and remission were 95.0%, 63.6% and 57.6%, respectively, at M12 post-dose escalation (Table). One case each of serious infection, herpes zoster (non-serious; opportunistic infection) and malignancy were reported (see Table).
Conclusion
For patients with moderate to severe UC who entered OLE in remission, received tofacitinib 5 mg BID but flared and were dose-escalated to 10 mg BID, response was recaptured for most by M3 post-dose-escalation and remission was recaptured in the majority by M12. Safety in this subpopulation was generally consistent with that observed in the overall tofacitinib UC programme.2 Due to the small sample size and open-label nature, results should be interpreted with caution.
Sandborn WJ
Sandborn WJ


