DOP84 Crohn’s disease modifies the DNA methylome of human adipose-stem cells, which is only partially re-established in remission
C. Serena1, M. Millan2, M. Ejarque1, A. Saera-Vila1, D. Monfort-Ferré1, M. Bautista3, M. Menacho3, M. Martí4, E. Espin4, J. Vendrell1, S. Fernández-Veledo1
1Health Institute Pere Virgili IISPV, Hospital Joan XXIII of Tarragona, Tarragona, Spain, 2General Surgery Service, Hospital Joan XXIII, Tarragona, Spain, 3Digestive Unit, Hospital Joan XXIIII, Tarragona, Spain, 4General Surgery Service, Hospital University Vall d’Hebron, Barcelona, Spain
Background
Crohn’s disease (CD) is characterised by the expansion of mesenteric adipose tissue, termed creeping fat (CF). We previously demonstrated that human adipose-tissue stem cells (hASCs) from CF exhibit a dysfunctional phenotype, including a pro-inflammatory profile, high phagocytic capacity and weak immunosuppressive properties. Importantly, these phenotypes persist in patients in remission, and are found in all adipose depots explored including subcutaneous fat. We hypothesised that this is a consequence of epigenetic modifications.
Methods
Epigenome-wide association studies using the 850k EPIC Illumina array were used to explore the impact of CD on the methylation signature of hASCs isolated from the subcutaneous fat (Figure 1). Changes in the expression of differentially methylated candidate genes were validated in hASCs of patients with active/inactive CD and healthy controls (Figure 1).
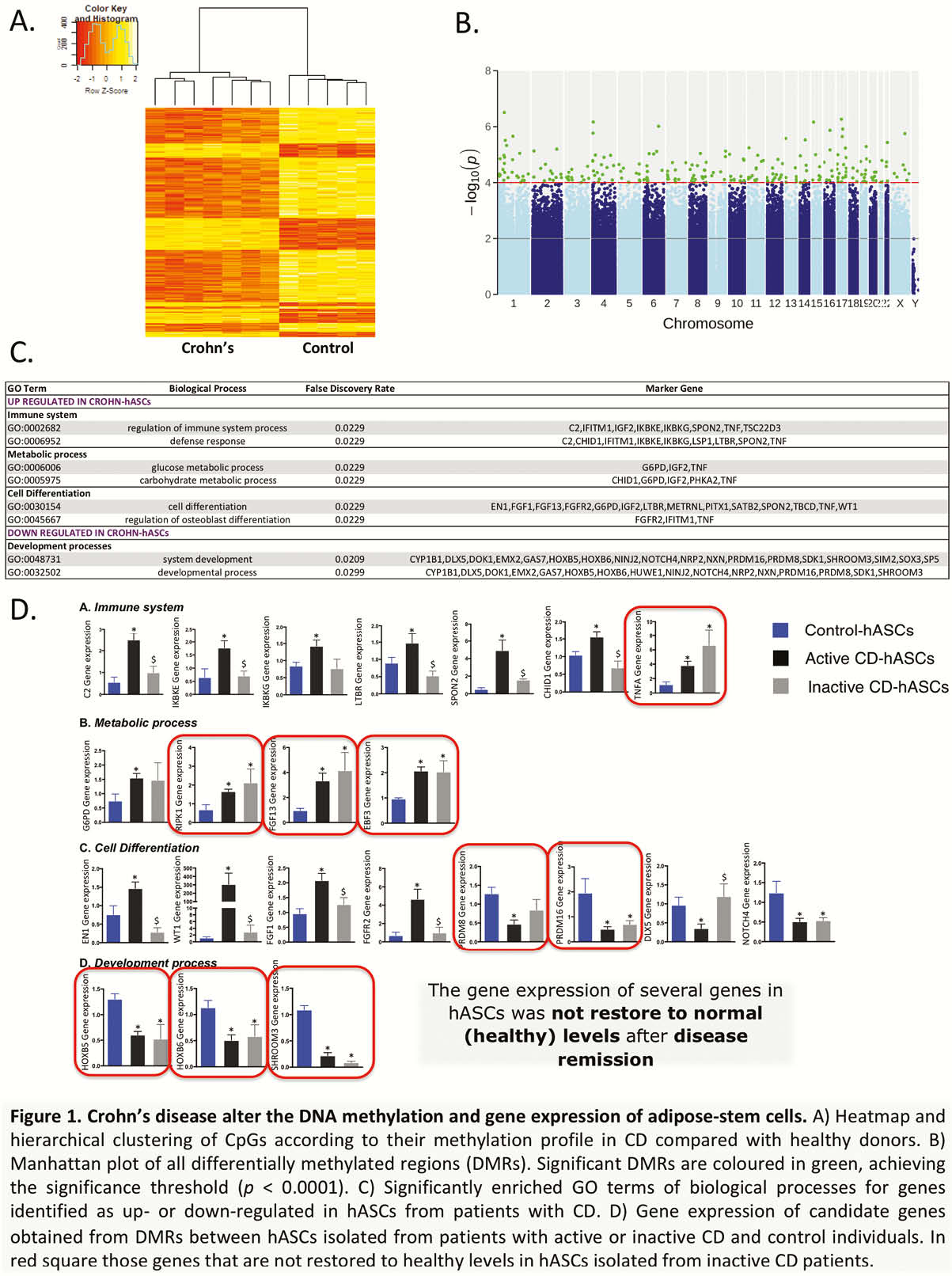
Results
hASCs of patients with CD exhibit an alter DNA-methylation and gene expression profile. Remarkably, the expression of several genes is only partially restored in patients with inactive CD. Understanding how CD shapes the functionality of hASCs is critical for investigating the complex pathophysiology of this disease, as well as for the success of cell-based therapies.


