DOP86 Corticosteroid-sparing effects of ustekinumab therapy for Ulcerative Colitis through 3 years: UNIFI long-term extension
Scherl, E.J.(1);Rowbotham, D.S.(2);Danese, S.(3);Sandborn, W.J.(4);Miao, Y.(5);Zhang, H.(5);Abreu, M.T.(6);Panaccione, R.(7);Afif, W.(8);Hisamatsu, T.(9);Leong, R.W.(10);Arasaradnam, R.P.(11);Sands, B.E.(12);Marano, C.(5)
(1)New York Presbyterian Weill Cornell Medical College, Roberts Center for Inflammatory Bowel Disease, New York, United States;(2)Auckland City Hospital, Gastroenterology, Auckland, New Zealand;(3)Instituto Clinico Humanitas, Department of Gastroenterology, Milano, Italy;(4)University of California San Diego, Division of Gastroenterology, La Jolla, United States;(5)Janssen Research & Development- LLC, Immunology, Spring House, United States;(6)University of Miami Miller School of Medicine, Department of Medicine, Miami, United States;(7)University of Calgary, Inflammatory Bowel Disease Unit, Calgary, Canada;(8)McGill University Health Centre, Division of Gastroenterology, Montreal, Canada;(9)Kyorin University School of Medicine, Third Department of Internal Medicine, Tokyo, Japan;(10)Concord Repatriation General Hospital, Inflammatory Bowel Disease Clinic, New South Wales, Australia;(11)University Hospital Coventry and Warwickshire, Gastrointestinal Medicine, Coventry, United Kingdom;(12)Mount Sinai School of Medicine, Henry D. Janowitz Division of Gastroenterology, New York, United States UNIFI
Background
Ustekinumab (UST) is an IL12/23 blocker approved for use in Crohn’s disease and ulcerative colitis (UC). In the UNIFI maintenance study of patients (pts) with moderate-severe UC, >90% of the pts who achieved clinical response or remission at week (wk) 44 were able to eliminate corticosteroids, an important goal of therapy. In this analysis, we describe the corticosteroid-sparing effects of UST treatment through 3 years among pts who were treated in the UNIFI long-term extension (LTE).
Methods
523 intravenous UST induction responders were randomized to SC maintenance therapy (SC placebo [PBO], n=175; UST 90mg every 12 wks [q12w], n=172; or UST 90mg q8w, n=176). 284 UST pts who completed wk44 entered the LTE. PBO pts were discontinued after wk44 unblinding. Based on the investigator’s clinical judgement of their UC disease activity, pts in the LTE were eligible to receive dose adjustment starting at wk56: PBO to q8w, q12w to q8w, and q8w to q8w (sham adjustment). Pts in PBO or q8w groups were only eligible for dose adjustment or sham dose adjustment before unblinding. Efficacy was evaluated in randomized pts using symptomatic remission (Mayo stool frequency subscore of 0 or 1 and a rectal bleeding subscore of 0).
During the maintenance study, all pts receiving corticosteroids at maintenance baseline were required to initiate tapering. Through wk152 of the LTE, symptomatic remission endpoints were calculated with treatment failure and missing data nonresponder imputation, and dose adjustment was not considered to be a treatment failure. Missing corticosteroid dose data was managed using last observation carried forward.
Results
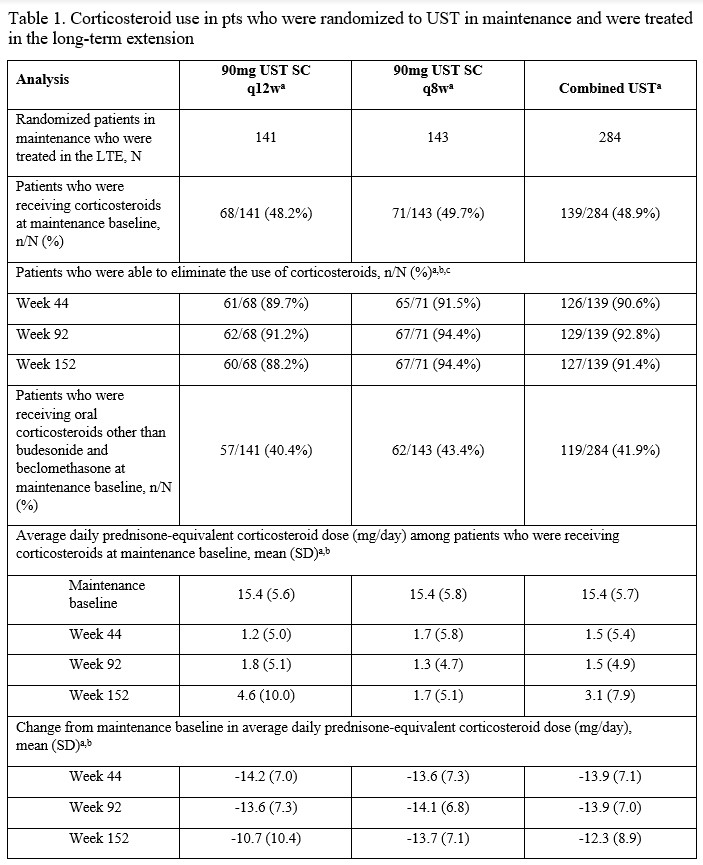
Of the 284 pts in the randomized population who were treated with UST in the LTE, 139 were receiving corticosteroids at maintenance baseline. Of these, 91.4% (n=127) were no longer receiving corticosteroids at wk152. The average prednisone-equivalent corticosteroid dose among pts receiving corticosteroids at maintenance baseline in the q8w group was 15.4 mg/day at maintenance baseline and 1.7 mg/day at wks44 & 152. In the q12w group, average prednisone-equivalent doses were 15.4, 1.0, and 4.6 mg/day, respectively (Table 1). Corticosteroid-free symptomatic remission rates through wk152 are summarized in Table 2. Results were similar for the q8w and q12w maintenance doses. Of the UST-treated pts who were in symptomatic remission at wk152, 94.6% (88/93) in the q12w group and 98.0% (97/99) in the q8w group were corticosteroid-free.
Conclusion
UST maintenance therapy, with both q8w and q12w dosing regimens, was effective in reducing and eliminating the use of corticosteroids in pts with UC through 3 years. Through 3 years of treatment with UST, the majority of pts in symptomatic remission were corticosteroid free.