OP34 Efficacy and safety of advanced induction and maintenance therapies in patients with moderately to severely active Ulcerative Colitis: An indirect treatment comparison using Bayesian network meta-analysis
Panaccione, R.(1);Collins, E.B.(2);Melmed, G.Y.(3);Vermeire, S.(4);Danese, S.(5);Higgins, P.D.R.(6);Zhou, W.(7);Ilo, D.(7);Sharma, D.(7);Sanchez Gonzalez, Y.(7);Wang, S.T.(2);
(1)University of Calgary, Inflammatory Bowel Disease Unit- Division of Gastroenterology and Hepatology, Calgary, Canada;(2)Medicus Economics LLC, n/a, Milton, United States;(3)Cedars-Sinai Medical Center, n/a, Los Angeles, United States;(4)University Hospital Leuven, Department of Gastroenterology and Hepatology, Leuven, Belgium;(5)Gastroenterology and Endoscopy IRCCS Ospedale San Raffaele and University Vita-Salute San Raffaele, n/a, Milan, Italy;(6)University of Michigan, Department of Medicine- Division of Gastroenterology, Ann Arbor, United States;(7)AbbVie Inc., n/a, North Chicago, United States;
Background
The therapeutic armamentarium to treat adult patients with moderately to severely active ulcerative colitis (UC) continues to evolve. With this rapid innovation, the comparative efficacy and safety of more recent advanced therapies remain unknown.
Methods
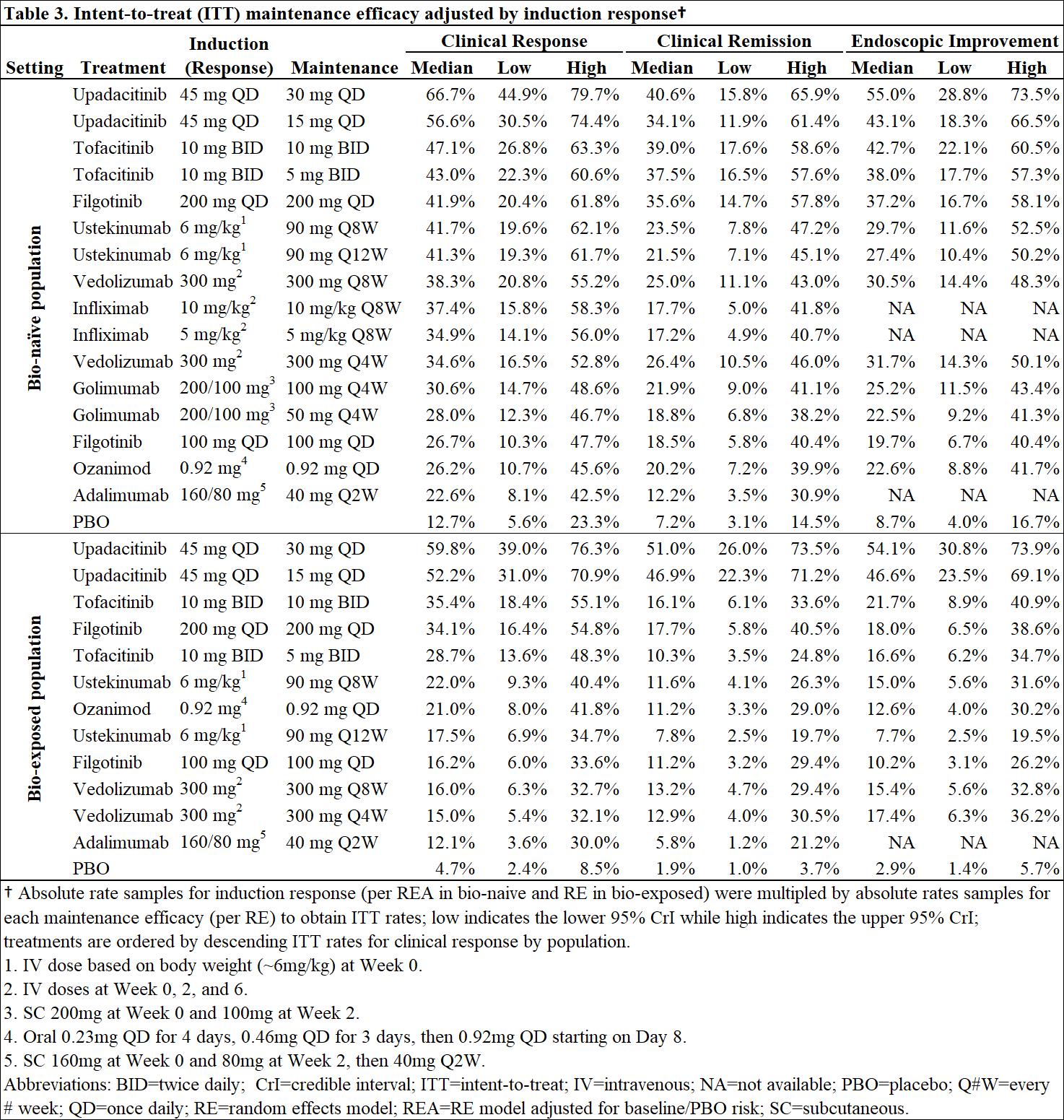
Bayesian network meta-analysis was used to indirectly compare the efficacy and safety of advanced therapies for induction (6-10 weeks) and maintenance (44-54 weeks post-induction response) in adults with moderately-to-severely active UC. Efficacy was assessed separately in bio-naïve and bio-exposed populations by clinical remission (Full Mayo score [FM] of ≤2 with no subscore >1), clinical response (decrease from baseline in FM ≥3 points and ≥30% with decrease in rectal bleeding score [RBS] of ≥1 or absolute RBS ≤1) and endoscopic improvement (endoscopic score ≤1); ad hoc analyses were conducted on upadacitinib (UPA) RCT data to produce FM outcomes. Safety was assessed by discontinuation due to adverse events (AEs), serious AEs, and serious infections. Induction therapies included UPA 45 mg, adalimumab 160/80 mg, filgotinib 100 and 200 mg, golimumab 200/100 mg, infliximab 10 and 5 mg/kg, ozanimod 0.92 mg, tofacitinib 10 mg, ustekinumab (UST) 6 mg/kg, and vedolizumab (VED) 300 mg. The maintenance analysis included low and high maintenance doses of these therapies. Phase 3 randomized controlled trials (RCTs) were identified via systematic literature review. Random effects models were used to account for expected heterogeneity in endpoints and study design. Guidelines from the National Institute for Health and Care Excellence were followed.
Results
Out of 31 RCTs identified, 23 were included (18 for induction and 14 for maintenance). Odds ratios vs. placebo (PBO), numbers needed to treat/harm, and surface under the cumulative ranking curve estimates are presented for efficacy in bio-naïve (Table 1) and bio-exposed (Table 2) populations and for safety in overall populations (Table 4). Intent-to-treat rates of maintenance efficacy outcomes adjusted by the likelihood of induction response show UPA to be consistently the most efficacious therapy (Table 3). There were no significant differences in serious AEs or serious infections for any advanced therapy vs PBO. For discontinuation due to AEs, only UPA had significantly lower odds vs PBO after induction, while UST and VED had significantly lower odds vs PBO after maintenance (Table 4).
Conclusion
In patients with moderately-to-severely active UC, UPA 45 mg induction and 30 mg maintenance appear more efficacious than other advanced therapies/PBO at inducing and maintaining clinical response, clinical remission, and endoscopic response, with no greater safety assessments vs PBO, over 1-year.


- Posted in: Scientific Session 10: Beyond the gut


