OP40 PRA023 Demonstrated Efficacy and Favorable Safety as Induction Therapy for Moderately to Severely Active UC: Phase 2 ARTEMIS-UC Study Results
B. Sands*(1), L. Peyrin-Biroulet(2), S. Danese(3), D.T. Rubin(4), S. Vermeire(5), O. Laurent(6), A. Luo(6), D. Nguyen(6), JD. Lu(6),
A. Wiechowska-Kozlowska(7), J. Leszczyszyn(8), R. Kempinski(9), J. Kierkus(10), C. Ma(11), T. Ritter*(12), B.G. Feagan(13), S. Targan*(14)
(1)Icahn School of Medicine at Mount Sinai, New York, New York, USA, (2)Last Inserm U954 and CHU de Nancy, Lorraine University, Nancy, France, (3)Gastroenterology and Gastrointestinal Endoscopy Unit, IRCCS San Raffaele Scientific Institute, Vita-Salute San Raffaele University, Milano, Italy, (4)University of Chicago Medicine Inflammatory Bowel Disease Center, Chicago, IL, USA, (5)University Hospitals Leuven,
Department of Gastroenterology and Hepatology, Leuven, Belgium, (6)Prometheus Biosciences, Inc., San Diego, CA, USA, (7)Endoscopy Unit, SONOMED Medical Center, Szczecin, Poland, (8)Department of Gastroenterology, Melita Medical, Wroclaw, Poland, (9)Wroclaw Medical University, Department of Gastroenterology and Hepatology, Wroclaw, Poland, (10)Department of Gastroenterology, Hepatology, Feeding Disorders and Pediatrics, The Children's Memorial Health Institute, Warsaw, Poland, (11)Alimentiv Inc., London, ON, Canada Division of Gastroenterology & Hepatology, Department of Medicine and Community Health Sciences, University of Calgary, Calgary, AB, Canada, (12)GI Alliance, Southlake, Texas, USA, (13)Department of Medicine, Division of Gastroenterology, Western University, London, ON, Canada, (14)Division of Gastroenterology and Hepatology, Center for Inflammatory Bowel Diseases, Cedars-Sinai Medical Center, Los Angeles, CA,USA
Background
Tumor necrosis factor-like cytokine 1A (TL1A) is an upstream regulator of pro-inflammatory cytokines and fibrosis signals. PRA023 is an anti-TL1A monoclonal antibody in development for multiple inflammatory/fibrotic diseases using a precision medicine approach with a companion diagnostic (CDx). This Phase 2 placebo-controlled, multi-center, double-blind study aimed to assess the efficacy and safety of PRA023 for induction treatment in adults with moderately to severely active ulcerative colitis (UC).
Methods
Key eligibility criteria included a modified Mayo Score of 4-9, a centrally read (with adjudication) endoscopy subscore of ≥ 2, a rectal bleeding subscore of ≥ 1, and a history of insufficient response, loss of response, and/or intolerance to conventional and/or advanced (approved biologics and JAK inhibitors/S1P modulators) therapies (≤ 4 advanced agents from ≤ 3 classes). Patients were stratified by prior biologic exposure status (yes/no) and CDx status (yes/no) and randomized 1:1 to placebo or intravenous PRA023 (1000mg on Day 1, 500mg at Weeks 2, 6, and 10). The primary endpoint was clinical remission at week 12. Analyses of all ranked secondary endpoints, the first being endoscopic improvement (endoscopy score of ≤ 1), were multiplicity controlled.
Results
Of the 135 patients in the full analysis data set, 60/67 (89.6%) in the placebo arm and 68/68 (100%) in the PRA023 arm completed the 12-week Induction Period. Baseline characteristics were similar with minor differences in the proportion of patients with baseline endoscopy score of 3 and prior exposure to ≥ 3 advanced therapies (Table 1). A significantly greater proportion of patients who received PRA023 achieved the primary endpoint of clinical remission (26.5% PRA023 vs. 1.5% placebo, ∆ 25.0%, p < 0.0001) at Week 12 (Table 2). In addition, substantially more patients who received PRA023 achieved the key secondary endpoint of endoscopic improvement (36.8% PRA023 vs. 6.0% placebo, ∆ 30.8%, p < 0.0001). All remaining ranked secondary endpoints were met (Table 2). The rates of treatment-emergent adverse events (AEs) were similar between the two arms with no serious AEs or AEs leading to study drug discontinuation in the PRA023 arm (Table 3). There were no safety signals identified from the study.
Conclusion
PRA023 was effective with favorable tolerability for the induction of clinical remission and endoscopic improvement in moderately to severely active UC. A Phase 3 study will be conducted to confirm these findings. Recruitment is ongoing for the CDx+ expansion cohort of the study to assess the effectiveness of the CDx, designed to select patients with a higher probability of responding to PRA023.
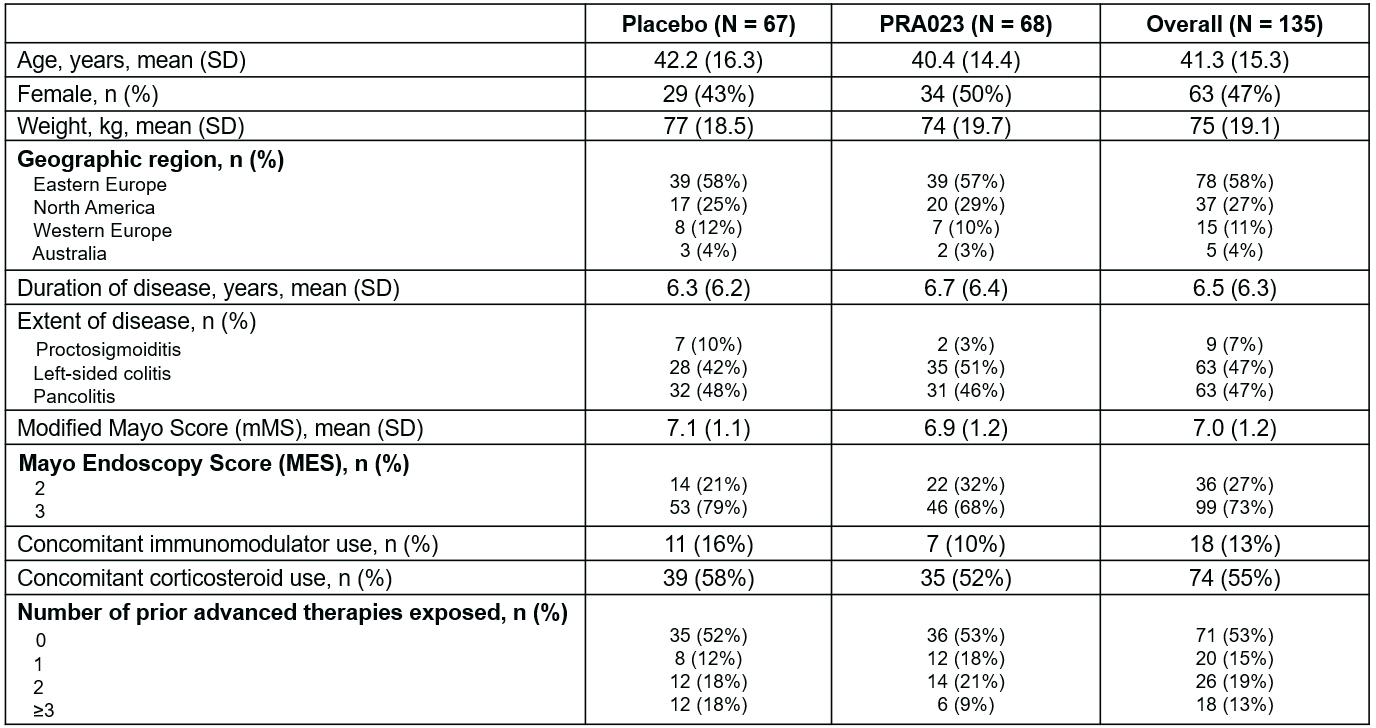
Table 1. Demographics and Baseline Characteristics.
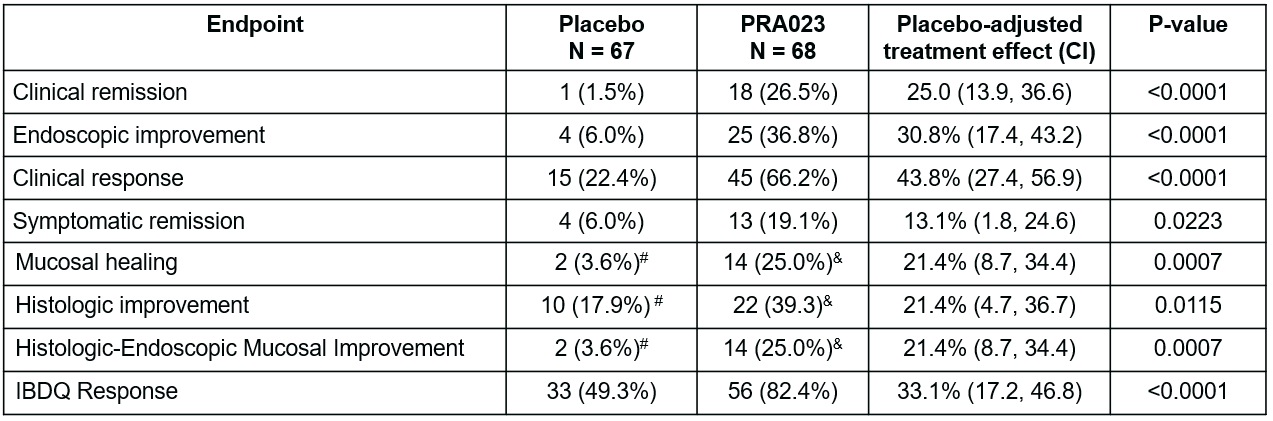
Table 2: Primary and Secondary Efficacy endpoints. Clinical remission per modified Mayo Score (mMS) is defined as endoscopic subscore of 0 or 1, rectal bleeding subscore of 0, and stool frequency subscore of 0 or 1 and not greater than Baseline. Endoscopic improvement is defined as endoscopy subscore ≤ 1 with no friability. Clinical response per mMS is defined as reduction from Baseline ≥ 2 points and ≥ 30% in mMS, accompanied by a reduction ≥ 1 in rectal bleeding subscore or absolute rectal bleeding subscore ≤ 1. Symptomatic remission is defined as stool frequency subscore of 0 and rectal bleeding subscore of 0. Histologic improvement is defined as Geboes score ≤ 3.1. Histologic-endoscopic mucosal improvement is defined as Geboes score ≤ 3.1 and endoscopy subscore ≤ 1; mucosal healing is defined as Geboes score ≤ 2B.1 and endoscopy subscore ≤ 1. IBDQ response is defined as IBDQ score increase of ≥ 16 points from Baseline. P-values for testing the treatment difference are based on Cochran-Mantel-Haenszel test adjusted for prior biologic exposure status and CDx status. All endpoints are statistically significant according to multiplicity controlled 2-sided alpha of 0.05. #n=56; &n=56. CI=confidence interval.
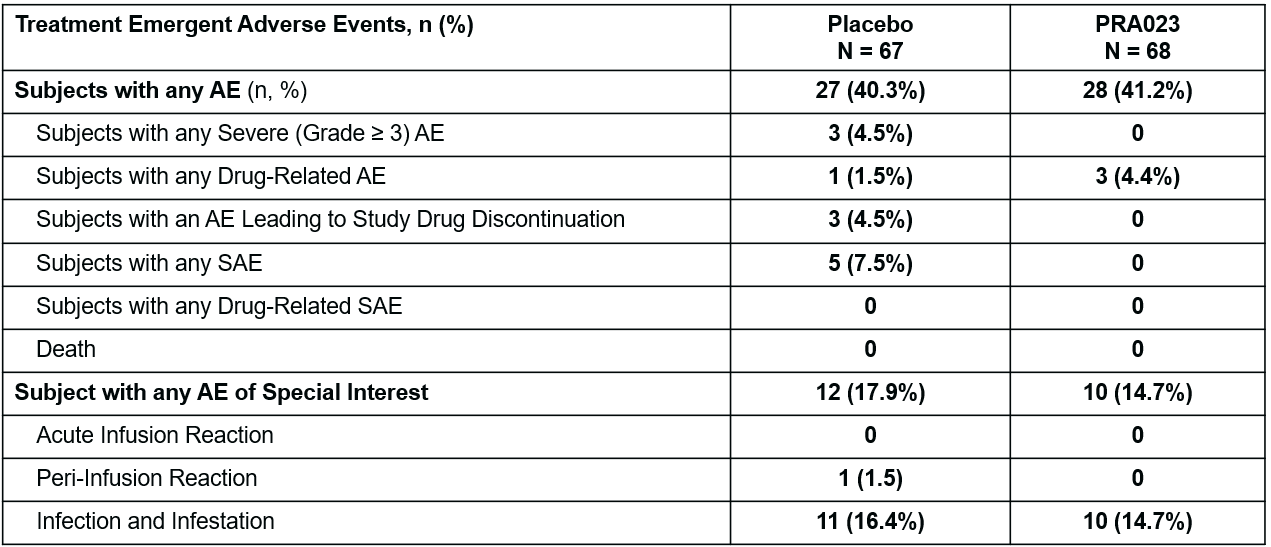
Table 3. Safety Summary Table. Acute infusion reaction: events as defined by the MedDRA hypersensitivity Standard MedDRA Queries (SMQ) occurring within 1 hour of completion of infusion. Peri-infusion reaction: events as defined by the MedDRA hypersensitivity SMQ occurring within 24 hours of completion of infusion.


