P019 Colonic mucosal kinase activity, cytokine and chemokine profiles in inflammatory bowel disease
E. Brand1, B. Roosenboom2, B. Malvar Fernandez3, L. Lutter1, E. van Koolwijk4, E.G. van Lochem4, C.S. Horjus Talabur Horje2, K.A. Reedquist3, F. van Wijk3, B. Oldenburg5
1Department of Gastroenterology and Hepatology and Centre for Translational Immunology, University Medical Centre Utrecht, Utrecht, The Netherlands, 2Department of Gastroenterology and Hepatology, Rijnstate Crohn and Colitis Centre, Arnhem, The Netherlands, 3Centre for Translational Immunology, University Medical Centre Utrecht, Utrecht, The Netherlands, 4Department of Microbiology and Immunology, Rijnstate Hospital, Arnhem, The Netherlands, 5Department of Gastroenterology and Hepatology, University Medical Centre Utrecht, Utrecht, The Netherlands
Background
With the approval of tofacitinib, an oral Janus Kinase (JAK) inhibitor, modulation of kinase activity has been added to the therapeutic armamentarium of inflammatory bowel disease (IBD). Despite its established efficacy, at least a third of patients will not respond to this or other therapeutic options such as anti-tumour necrosis factor (TNF), anti-interleukin (IL)23/IL12 compounds or vedolizumab. A better understanding of the inflammatory profile could aid in tailoring drugs to individual patients. We therefore explored mucosal cytokine, chemokine and kinase activity profiles in IBD.
Methods
Colonic mucosal biopsies were collected from (1) patients with Crohn’s disease (CD,
Results
Whole-biopsy kinase activity differed between inflamed and non-inflamed mucosa of IBD patients, with more overall tyrosine kinase activity in inflamed mucosa in UC, and serine/threonine kinase activity in inflamed mucosa in CD as compared with non-inflamed mucosa (Figure 1). The kinase activity profile of non-inflamed mucosa of CD and UC patients was similarly different from mucosa of healthy control participants (Figure 2). The cytokine and chemokine profile of inflamed biopsies differed from non-inflamed IBD biopsies and healthy control biopsies, with higher levels of S100A8, TNFα, IL-6, oncostatin M (OSM) and triggering receptor expressed on myeloid cells-1 (TREM-1), amongst others (Figure 3).
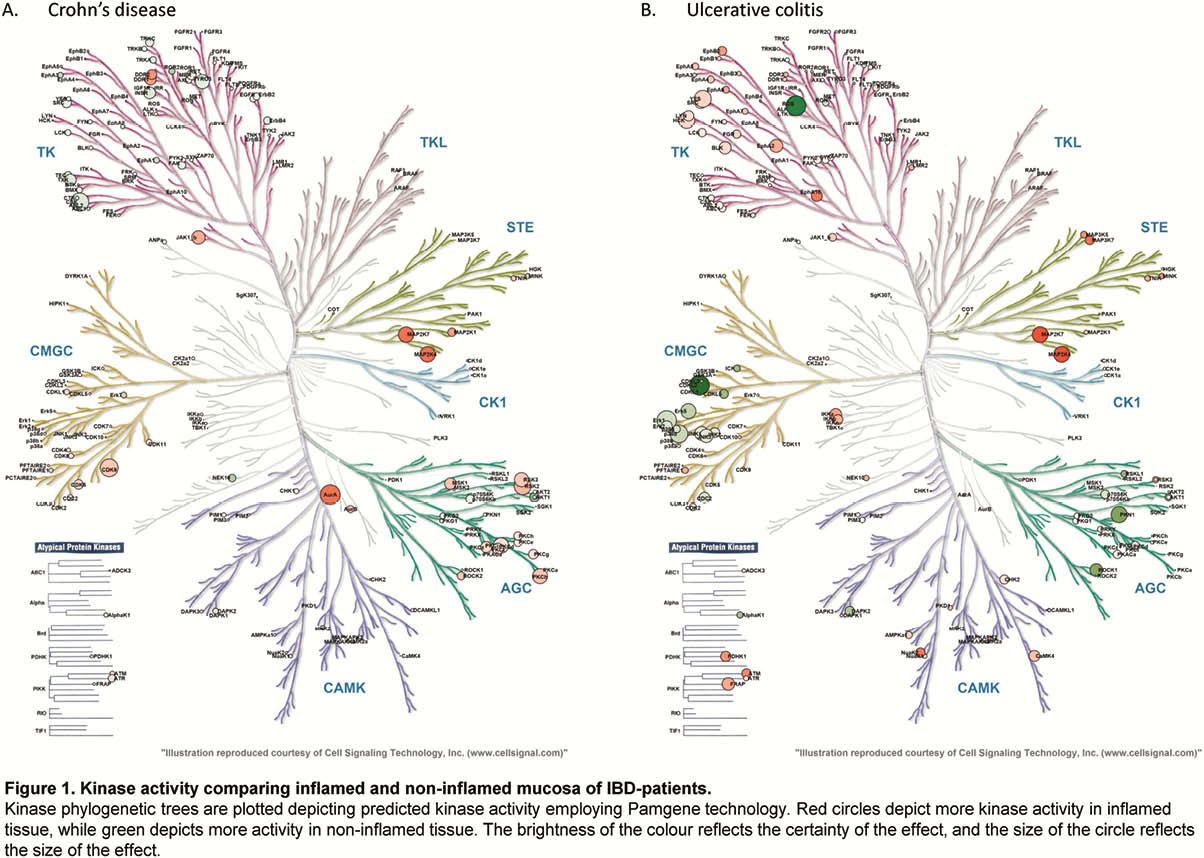
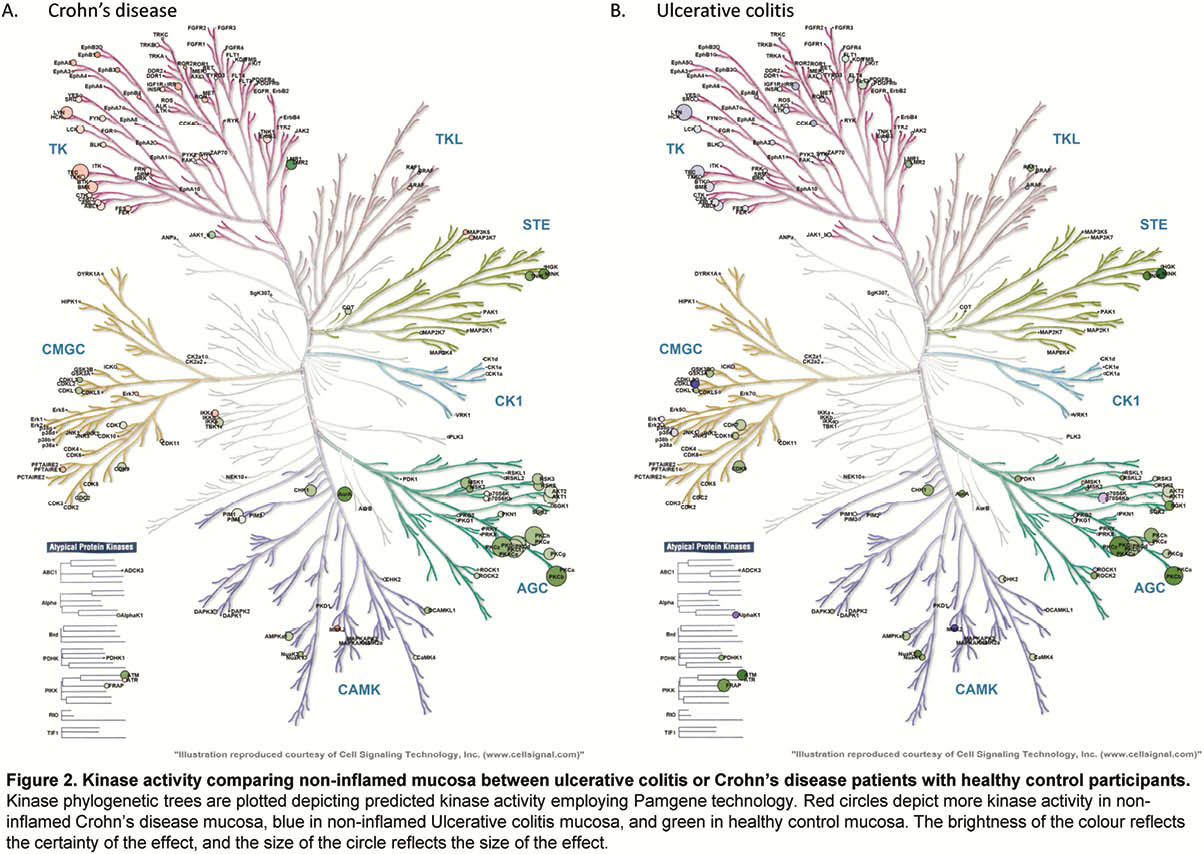
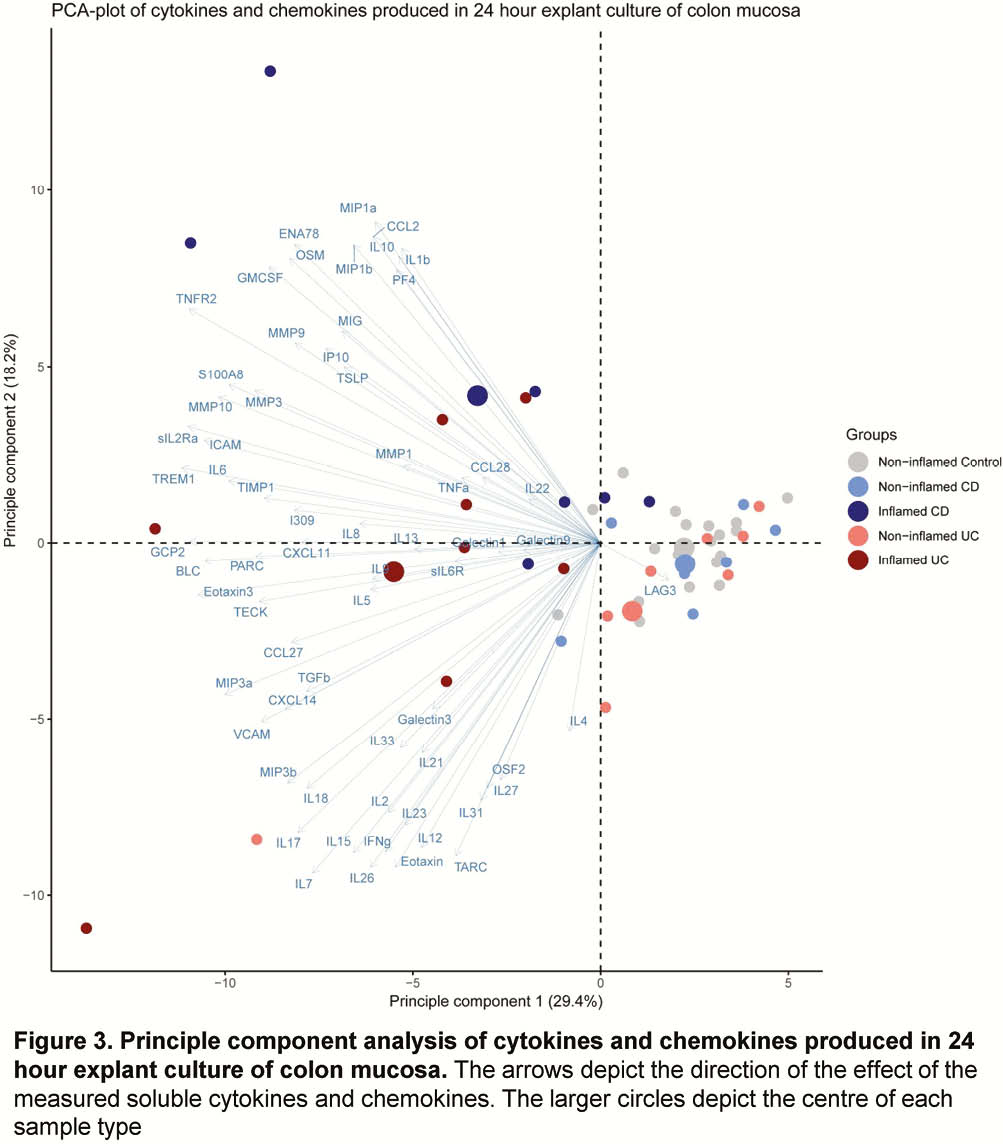
Conclusion
In IBD, inflammation in the mucosa can be characterised both by explant-culture and kinase activity assessment. The difference in kinase activity between non-inflamed IBD mucosa and healthy control mucosa suggests the presence of sub-clinical alterations in cell signalling. The observed differences in the kinase, cytokine and chemokine profiles underscore the importance of this approach in the elucidation of the pathophysiology in IBD.


