P045 Local administration of extracellular vesicles from adipose-derived stromal cells promote healing in a rat model of radiation colitis – a pilot study
Sebbagh, A.(1)*;Mathieu, N.(2);Demarquay, C.(2);Geiger, M.(2);Sémont, A.(2);Simon, C.(3);Branchu, J.(3);Milliat, F.(2);Gazeau, F.(1);Wilhelm, C.(4);Silva Brun, A.(1);Rahmi, G.(5,6);
(1)Laboratoire Matière et Systèmes Complexes UMR 7057- Université Paris Cité/CNRS, MSC-Med, Paris, France;(2)Institut de radioprotection et de sûreté Nucléaire IRSN, PSE-SANTE/SERAMED/LRMed, Fontenay-aux-Roses, France;(3)EVerZom company, Internal research and development, Paris, France;(4)Institut Curie, Physico-Chimie Curie CNRS UMR 168, Paris, France;(5)Hôpital Européen Georges Pompidou- APHP.Centre – Université de Paris, Department of Gastroenterology and Endoscopy, Paris, France;(6)Paris Cardiovascular Research Center, Laboratoire Imagerie de l’Angiogénèse- Plateforme d’Imagerie du Petit Animal- Inserm UMR 970, Paris, France;
Background
The estimated increase in prevalence of radiation enteropathy associated with a current scarcity of effective treatment options underline the need for the development of new therapies. Extracellular vesicles (EV), which are membrane-bound particles secreted by cells containing proteins, lipids and nucleic acids, have yielded promising preclinical results in various applications in regenerative medicine. Here, we aimed to investigate whether EV from adipose-derived stromal cells (ADSC) may present a therapeutic potential in the context of radiation colitis.
Methods
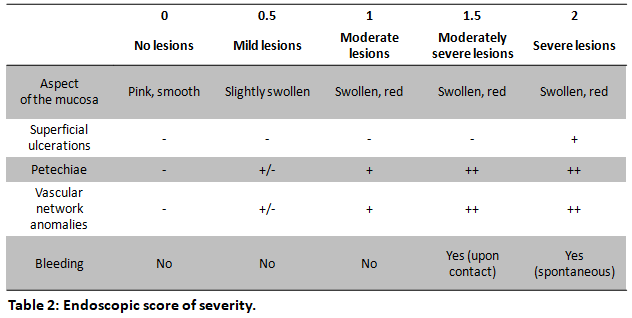
EV were produced from primary rat ADSC by exposure to a turbulent flow. Radiation colitis was induced by fractionated irradiation at a dose of 3x10Gy on a colorectal window (3x2cm) in male Sprague-Dawley rats (311-386g) at days 0, 2 and 4; following a pre-established protocol. Treatment was administered at days 11 and 15 through submucosal endoscopic injections of rat ADSC (n=9 rats, 2x106cells per dose), EV (n=9 rats, 1.06x1011 particles per dose), or an equivalent volume of saline (n=10 rats). The rats were sacrificed at day 17. Our primary endpoint was the extent of colonic epithelial injury at histological analysis (Table 1). Secondary endpoints included an endoscopic assessment of the severity of lesions at day 15 (Table 2) and an evaluation of macrophage infiltration in the colonic mucosa by CD68 immunohistochemistry. Comparisons of proportions were performed by the Chi-squared test followed by Fisher’s exact test.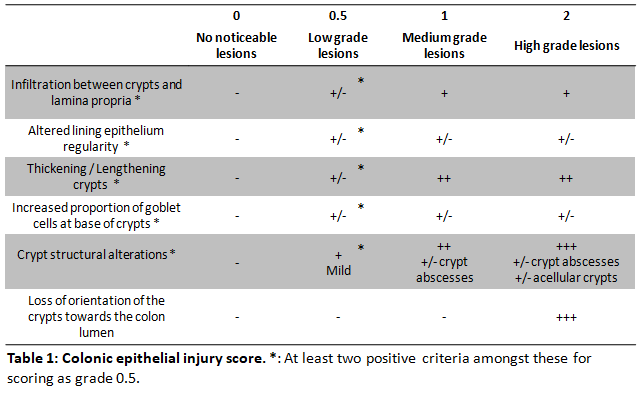
Results
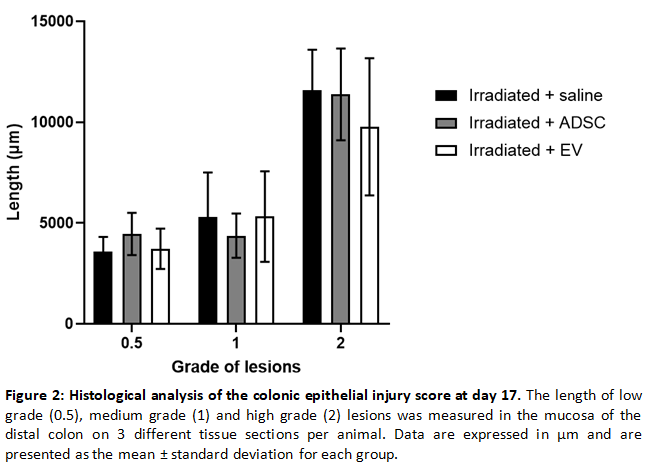
In the ADSC group, 1 rat died from anaesthesia complications. No other adverse effects were observed. No differences in total body weight evolution were observed between the groups. At day 15, a statistically significant decrease of the proportion of animals with moderate to severe lesions was evidenced by endoscopy in the EV group compared to the saline group (p=0.0031; Figure 1). No differences were observed between the other groups. Histological analysis of the colonic epithelial injury score evidenced a non-significant tendency towards a decreased length of high-grade lesions in the EV group (mean±standard deviation: 9180±3684µm; Figure 2) compared with the saline (11536±2711µm) and ADSC (11636±2815) groups. Similarly, CD68+ macrophage infiltration at the site of colonic mucosal lesions tended to be lower in the EV and ADSC groups compared to the saline group.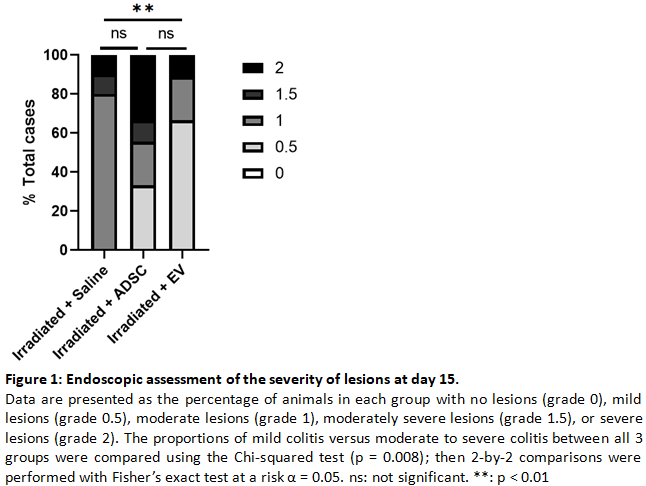
Conclusion
This proof-of-concept study seemed to demonstrate a beneficial effect of locally administered extracellular vesicles for the treatment of radiation colitis in the acute setting. A confirmation study in a larger animal cohort is underway, and further investigations focusing on dose effects or long-term effects on fibrosis are required to complete these preliminary results.


