P045 Profiling of human circulating dendritic cells and monocytes subsets discriminates type and mucosal status in patients with inflammatory bowel disease
L. Ortega Moreno1, S. Fernández Tomé1, M. Chaparro1, A. Marin1, I. Mora Gutiérrez1, C. Santander1, M. Baldán1, J. Gisbert1, D. Bernardo2
1Hospital Universitario de La Princesa, Medicina, Madrid, Spain, 2Universidad de Valladolid, Instituto de Biología y Genética Molecular, Valladolid, Spain
Background
Intestinal dendritic cells (DCs) and macrophages govern the mechanisms of immune homeostasis having a role in inflammatory bowel disease (IBD) onset. However, the profile of their circulating precursors (DC and monocytes) in IBD has not been previously described in depth. Our aim was to characterise blood DC and monocyte subsets in healthy controls (HCs) and IBD patients in order to understand their potential implication in IBD pathogenesis.
Methods
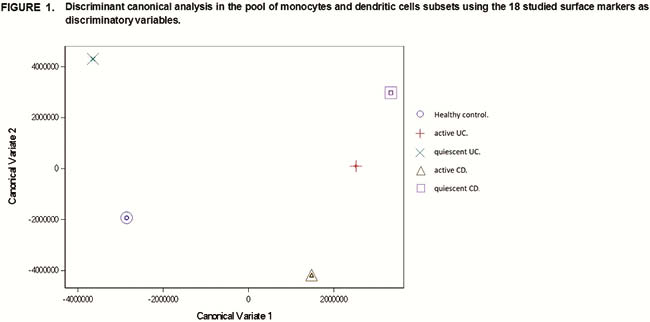
18 HC and 64 IBD patients were recruited. IBD patients were categorised into Crohn’s disease (CD) and ulcerative colitis (UC), either endoscopically active (aCD and aUC) or quiescent (qCD and qUC), based on the SES-CD or the Mayo index endoscopic subscore. Blood circulating type 1 conventional DC (cDC1), type 2 conventional DC (cDC2), plasmacytoid DC (pDC), classical monocytes, non-classical monocytes and intermediate monocytes were identified by flow cytometry and characterised for the expression of 18 homing and activation markers (β7, CCR1, CCR2, CCR3, CCR5, CCR6, CCR7, CCR9, CCRL1, CD40, CD86, CD137L, CD274 (PD-L1), CLA, CXCR1, CXCR3, ICOSL and HLA-DR). Association between markers and the presence, type or activity of IBD was tested by logistic regression. Discriminant canonical analysis was also performed to classify the patients on their own endoscopy category.
Results
All groups (HC, aCD, qCD, aUC and qUC) were separated from the others based on the discriminant canonical analyses of the 18 markers applied over all DC and monocytes subsets (Figure 1). Specifically, CCRL1, CCR3 and CCR5 expression on cDC1, CCRL1 on non-classical monocytes and CCR9 and b7 on classical monocytes were highly associated to IBD. CCR3 displayed an odds ratio (OR) of 2.29 along with its 95% confidential interval (CI) between 1.11 and 4.75, showing a strong association with activity in CD; whereas the other markers displayed an inverse association with IBD. Hence, expression of CCRL1 on cDC1 and non-classical monocytes from aUC showed an OR (95% CI) of 0.23 (0.08–0.66) and 0.52 (0.28–0.95), respectively. In the case of qUC, CCR5 on cDC1 and β7 on classical monocytes displayed an OR (95% CI) of 0.10 (0.01–0.83) and 0.56 (0.34–0.90), respectively. CCR9 was inversely associated to qCD with an OR (95% CI) of 0.64 (0.46–0.89) in the classical monocytes subset. Indeed, the same markers (excluding β7) were also associated with IBD when all DC and monocyte subsets were considered at the same time.
Conclusion
Differences on the expression of migration markers CCR3, CCR5, CCR9, b7 and decoy receptor CCRL1 on circulating DC and monocyte subsets from IBD groups suggest the presence of constitutive migratory differences underlying IBD pathogenesis in CD or UC and its condition (inflamed or non-inflamed).


