P056 Kefir (Lactobacillus kefiranofaciens) promotes autophagy and protects the intestinal barrier against DSS-induced colitis
K.N. Da Silva1, T.S. Cordeiro1, A.G. Fávero1, C.S. Bogsan2, R.D.C. Sinigaglia3, S.J. Miszputen1, O. Ambrogini–Júnior1, A.P.R. Paiotti1
1Universidade Federal de São Paulo – Escola Paulista de Medicina UNIFESP/EPM, Discipline of Gastroenterology, Sao Paulo, Brazil, 2Universidade de São Paulo – USP, Faculty of Pharmaceutical Sciences, São Paulo, Brazil, 3Universidade Federal de São Paulo – Escola Paulista de Medicina UNIFESP/EPM, Electron Microscopy Center, Sao Paulo, Brazil
Background
Inflammatory bowel disease occurs when luminal antigens from microbiota stimulate the immune system of an intestinal barrier through defects in the tight junctions lead to an exacerbated response in the genetically predisposed individual. It is well known that the consumption of probiotics improves intestinal health. Specifically, the
Methods
Forty male Wistar rats were randomised into five groups: Sham; 5% DSS-induced colitis; control Kefir; 5% DSS-induced colitis treated with Kefir; 5% DSS-induced colitis treated with Kefir + 5-ASA (125mg/kg/day) (Ethics Committee on the Use of Animals number 3627290119). The lactic culture of Kefir (Danisco Biolacta, Olstyn, Poland) was administered
Results
Kefir treatment reduces the severity of DSS-induced colitis evidenced by decreased abdominal discomfort and rectal bleeding. Histologically, there was a significant decreased of colonic damage and preservation of goblet cells (
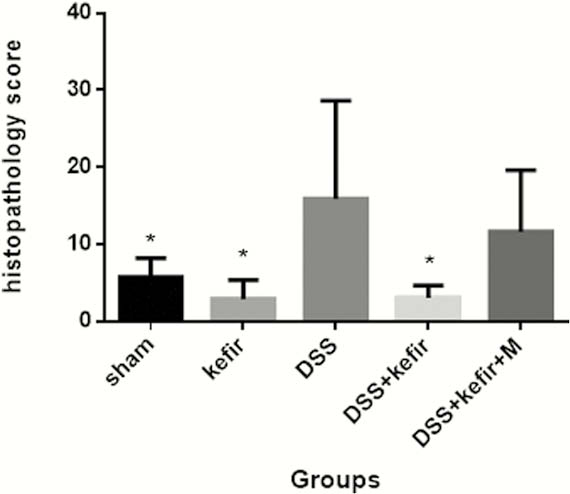
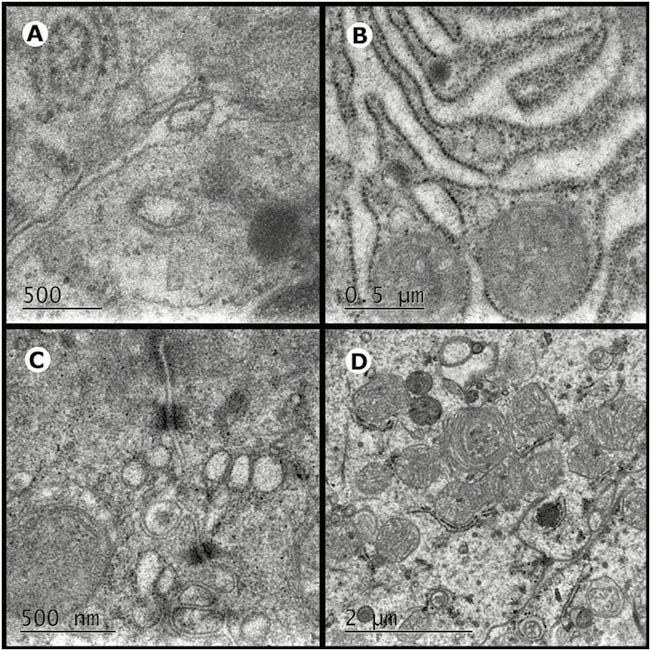
Conclusion
Kefir promotes autophagy and protects the intestinal barrier against damage caused by DSS.
- Posted in: Poster presentations: Basic Science 2020


