P091 Dietary triggers of colonic inflammation following treatment with exclusive enteral nutrition in children with Crohn’s disease
K. Gkikas1, M. Logan1, B. Nichols1, C.M. Clark1, U.Z. Ijaz2, L. Gervais3, H. Duncan3, V. Garrick3, L. Curtis3, E. Buchanan3, T. Cardigan3, L. Armstrong4, C. Delahunty5, D.M. Flynn3, A.R. Barclay3, R. Tayler3, R. Hansen3, R.K. Russell3, K. Gerasimidis1
1University of Glasgow, School of Medicine, Glasgow, UK, 2University of Glasgow, School of Engineering, Glasgow, UK, 3Royal Hospital for Children, Paediatric Gastroenterology, Hepatology and Nutrition, Glasgow, UK, 4Crosshouse Hospital, Department of Paediatrics, Kilmarnock, UK, 5Wishaw General Hospital, Department of Paediatrics, Wishaw, UK
Background
Exclusive enteral nutrition (EEN) ameliorates gut inflammation in children with Crohn’s disease (CD). We have previously described the rapid rise in faecal calprotectin levels (FC) when children with CD return to their habitual diet after EEN treatment (1). We performed dietary assessment and measured biomarkers of food consumption in faeces of children with CD during early food reintroduction.
Methods
A composite outcome of clinical remission post EEN (weighted Paediatric Crohn’s Disease Activity Index <12.5) and a significant drop in FC (FC decrease: >500 mg/kg / >35%) was used to define the patient group. All patients completed 3-day estimated weight food diaries and provided a faecal sample. Patients were divided equally for statistical analysis purposes in two groups; above (Group 1) and below (Group 2) the median FC concentration at food reintroduction. Nutrient analysis was performed with WinDiets. Short-chain fatty acids, the gluten immunogenic peptide (GIP) and starch were measured in stool, as proxy of fibre, gluten and malabsorbed starch, respectively. Food groups were assigned using the National Diet and Nutrition Survey approach and statistics employing taxonomy were utilised in the food group analysis. All data are displayed using medians (Q1, Q3).
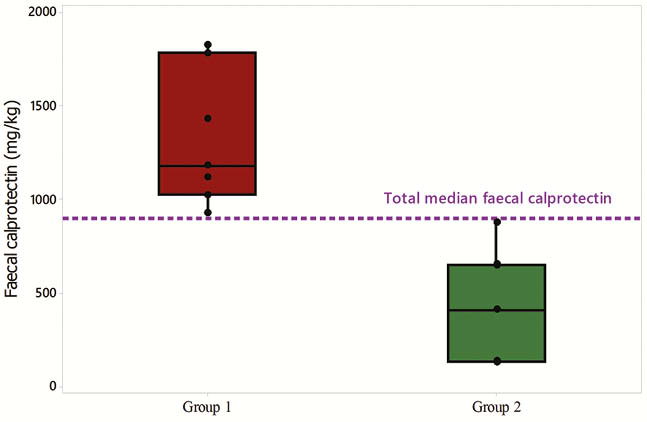
Results
Fourteen children provided an FC sample within 21 (15, 51) days post EEN. Patients were of similar age, weight and height (
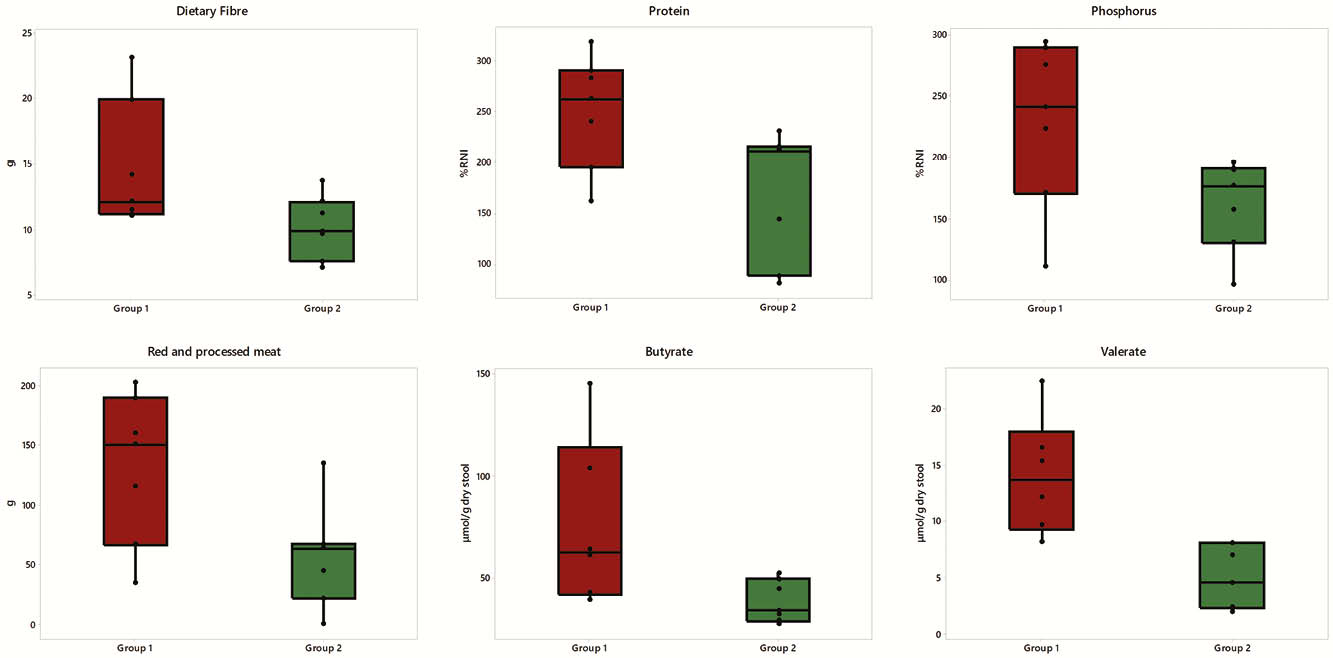
Conclusion
The current analysis suggests that fibre, protein, phosphorus and red and processed meat may be associated with recurrence of colonic inflammation in children with CD during early food reintroduction. These findings should be confirmed in larger studies.
Logan et al., Aliment Pharmacol Ther 50, 664–674, 2019


