P176 Expert consensus on acquisition and reporting of intestinal ultrasonography activity in Crohn’s disease. A prospective inter-rater agreement study
R.T. Wilkens1, K. Nylund2, F. Petersen3, F. De Voogd4, C. Maaser5, T. Kucharzik3, G. Maconi6, M. Allocca7, C. Lu8, C. Palmela9, R. Vaughan10, B. Christensen10, K.L. Novak8
1IBUS studygroup, 1Hvidovre Hospital-, Gastrounit- Division of Medicine, Copenhagen, Denmark, 2Haukeland University Hospital, Department of medicine, Bergen, Norway, 3University Teaching Hospital Lueneburg., Department of Internal Medicine and Gastroenterology-, Lueneburg, Germany, 4Amsterdam University Medical Centers, Department of Gastroenterology and Hepatology, Amsterdam, The Netherlands, 5University Teaching Hospital Lueneburg, Outpatients Department of Gastroenterology, Lueneburg, Germany, 6’Luigi Sacco’ University Hospital- University of Milan, Gastroenterology Unit- Department of Biomedical and Clinical Sciences, Milan, Italy, 7Humanitas University, Department of Gastroenterology, Milan, Italy, 8University of Calgary, Department of Medicine- Division of Gastroenterology, Calgary, Canada, 9Hospital Beatriz Ângelo, Gastroenterology Department, Loures, Portugal, 10The Royal Melbourne Hospital, Gastroenterology Department, Melbourne, Australia
Background
Intestinal ultrasonography (IUS) is a promising cross-sectional imaging modality used to assess transmural disease and complications in Crohn’s disease (CD). Although recently positioned as a first-line modality for evaluation as per ECCO guidelines, standard measurements, reproducibility and nomenclature have not yet been clearly established. The aim of this study was to evaluate the inter-rater agreement for parameters identified as important by experts through Delphi consensus.
Methods
IUS parameters demonstrating inflammatory activity were systematically reviewed in the literature and presented to IUS experts. Individual parameters were selected by a blinded Delphi consensus panel to establish relative contribution to inflammatory activity in CD. Weighted grading of each parameter was further established by expert consensus. Image acquisition for optimal measurement was established by consensus. Two phases for evaluating inter-rater variability were undertaken. Phase 1: blind review by 8 readers of 20 de-identified CD cases. Cases with poor agreement were reviewed to clarify discrepancy and improve agreement. Phase 2: an additional 30 de-identified CD cases blindly were reviewed by 12 independent expert readers. Inter-rater agreement was evaluated for all 4 key parameters. Statistics were performed using Stata 16. Bowel wall thickness (BWT) was assessed using intraclass correlation coefficient (ICC) and the ordinal parameters using weighted Cohens Kappa.
Results
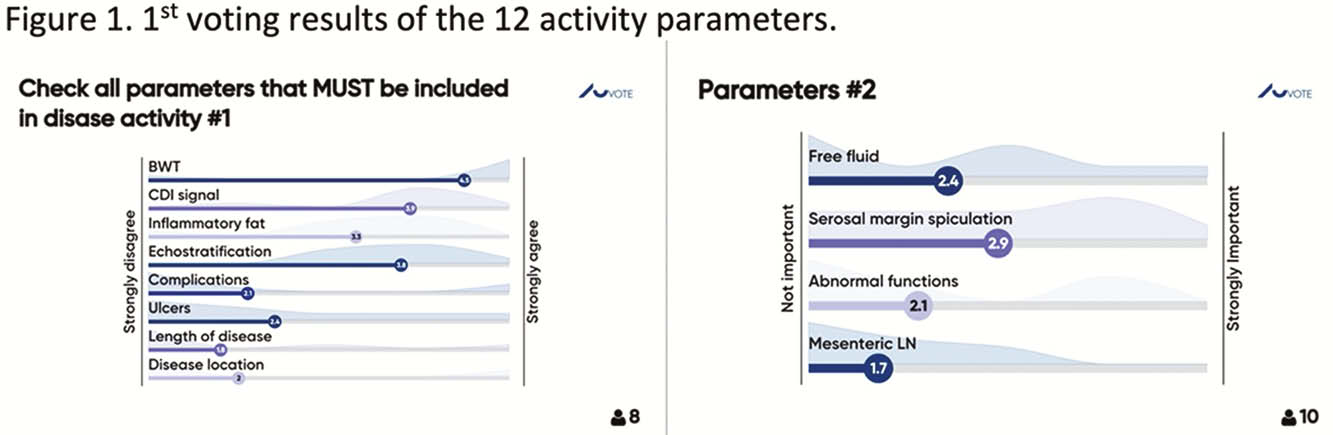
The Delphi process reduced 12 activity parameters to 4 key contributors including BWT, color Doppler signal (CDI), inflammatory fat and bowel wall echostratification (Figure 1).
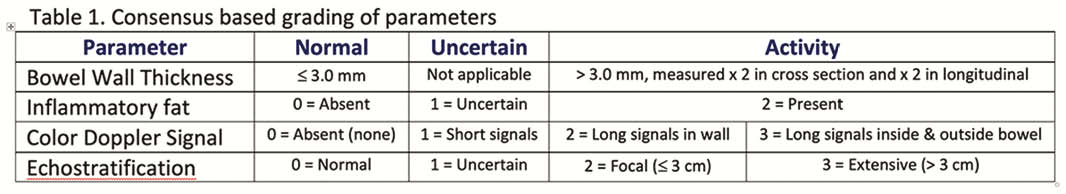
BWT was regarded as pathologic if the average of 4 measurements were > 3 mm for the small and large bowel, and grades of the additional parameters established (Table 1).
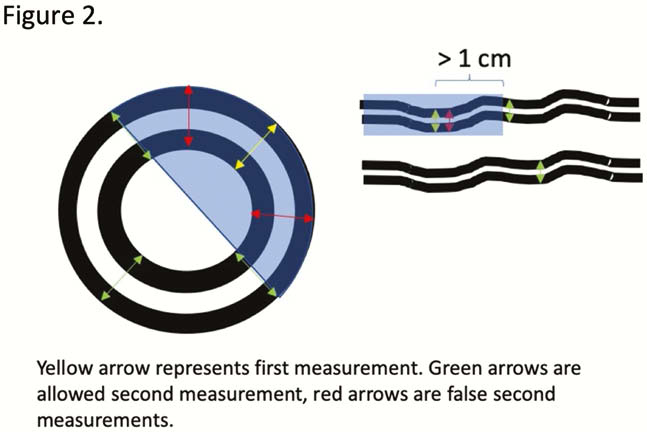
Bowel wall thickness was comprised of 2 measurements in cross section and 2 in longitudinal orientation (Figure 2). Interobserver agreement was almost perfect for BWT: ICC=0.91 (95% CI 0.83 to 0.96)
Conclusion
This expert consensus-based IUS activity score clearly establishes the reproducibility of this standardised approach to measure inflammatory activity in patients with CD. Using our method, BWT which is known as the most important parameter, is highly reproducible with CDI and inflammatory fat demonstrating moderate reproducibility. This score may provide the foundation for the future incorporation of IUS in research studies and clinical trials.


