Revés, J.(1);Morão, B.(1);Gomes, C.F.(1);Nascimento, C.(1);Abreu, N.(2);Gonçalves, T.C.(3);Castro, F.(3);Freitas, M.(3);Moreira, M.J.(3);Cotter, J.(3);Pereira, F.(4);Caldeira, A.(4);Sousa, R.(4);Coelho, R.(5);Macedo, G.(5);Macedo, C.(6);Ferreira, M.(6);Glória, L.(1);Torres, J.(1);Palmela, C.(1);
(1)Hospital Beatriz Ângelo, Gastrenterology Department, Loures, Portugal;(2)Hospital Dr. Nélio Mendonça, Gastrenterology Department, Funchal, Portugal;(3)Hospital da Senhora da Oliveira- Life and Health Sciences Research Institute- School of Medicine- University of Minho- Braga- ICVS/3B’s- PT Government Associate Laboratory, Gastrenterology Department, Guimarães/Braga, Portugal;(4)Unidade Local de Saúde de Castelo Branco- EPE- Hospital Amato Lusitano, Gastrenterology Department, Castelo Branco, Portugal;(5)Centro Hospitalar de São João- Porto, Gastrenterology Department, Porto, Portugal;(6)Centro Hospitalar Universitário de Coimbra, Gastrenterology Department, Coimbra, Portugal;
Background
Faecal calprotectin (FCal) is considered an intermediate target for monitoring disease activity in IBD. However, it is unknown whether early FCal variations during induction therapy may be a significant predictor of treatment response. We aimed to investigate FCal variation after infliximab induction therapy and its association with clinical, endoscopic, and ultrasonographic remission by the end of one year of treatment.
Methods
Prospective multicentric cohort study including patients with active CD without previous intestinal surgeries, who were starting IFX therapy and were followed for 54 weeks. FCal was measured at week 0 and after induction therapy (week 14) and relative FCal changes from baseline (%FCal0-14) were calculated. At week 54, clinical remission (defined as Harvey-Bradshaw index (HBI) <5), endoscopic healing (defined as SES-CD score <3) and transmural healing (defined as bowel wall thickness (BWT) assessed by intestinal ultrasound (IUS) ≤3mm) were evaluated.
Results
We included 33 patients (60.6% male; median age 30 years old (IQR 24-42)). Most patients were diagnosed between 17-40 years old (75.8%) had ileocolonic disease (57.6%) and an inflammatory phenotype (57.6%). At week 54, 90.9% of the patients were in clinical remission, 30.3% had endoscopic healing and 39.4% had transmural healing. The median FCal value at week 0 was 765µg/g (IQR 312-1313) and at week 14 was 124.5µg/g (IQR 46-432.5). At week 14, 20 patients (60.6%) had FCal<250µg/g. There was no correlation between FCal at week 0 and the baseline total SES-CD (r=0.28, p=0.13) and BWT of the most affected segment (r=0.02, p=0.90). Patients who achieved clinical remission had a low variation in %FCal
0-14 (87.4% vs 80.1%, p=0.42), as opposed to patients who achieved endoscopic healing (58.5 vs 91.3%, p<0.001) and transmural healing (68.6% vs 84.4%, p=0.16) who had more significant changes in %FCal
0-14, although only statistically significant for endoscopic healing. When stratifying %FCal
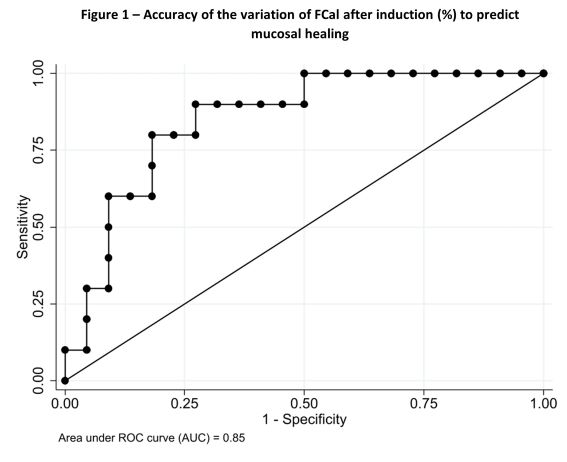
0-14 changes in 25% quartiles, FCal variation at the end of induction was only able to predict endoscopic healing (OR 8.13 (95%CI 1.01-65.46), p=0.049), being non-significant for clinical remission (OR 0.51 (95%CI 0.09-2.92), p=0.45) and transmural healing (OR 1.61 (95%CI 0.73-3.57), p=0.24). A normalization of FCal below 250 µg/g was also a predictor of endoscopic healing (OR 9.82 (95%CI 1.06-90.59), p=0.04). The AUC for the prediction of endoscopic healing with %FCal
0-14 changes was 0.85 (Figure 1) and a cut-off of variation of 80.3% was calculated.
Conclusion
FCal variation at the end of induction seems to be a good predictor of endoscopic healing at week 54 but does not seem to be able to predict clinical and ultrasonographic remission.



