P448 Oral beclomethasone dipropionate as an add-on therapy and response prediction in Korean patients with ulcerative colitis
Kim, K.(1);Hong, H.S.(1);Oh, K.(1);Lee, J.Y.(1);Hong, S.W.(1);Park, J.H.(1);Hwang, S.W.(1,2);Yang, D.H.(1);Byeon, J.S.(1);Ye, B.D.(1,2);Myung, S.J.(1);Yang, S.K.(1,2);Park, S.H.(1,2);
(1)University of Ulsan College of Medicine- Asan Medical Center, Department of Gastroenterology, Seoul, Korea- Republic Of;(2)University of Ulsan College of Medicine- Asan Medical Center, Inflammatory Bowel Disease Center, Seoul, Korea- Republic Of;
Background
We aimed to investigate the efficacy of oral beclomethasone dipropionate (BDP) as an add-on therapy to concomitant drugs and to clarify the predictive factors for response to oral BDP in Korean patients with ulcerative colitis (UC).
Methods
Patients with a stable concomitant drug regimen with exposure to oral BDP (5 mg/day) within 30 days before initiation were included. The partial Mayo score (pMS) was used to evaluate response to oral BDP. Clinical remission (CREM) was defined as a posttreatment pMS ≤1 point. Clinical response (CRES) was defined as an at least 2-points decrease in the posttreatment pMS and an at least 30% decrease from the baseline pMS. Patients without CREM or CRES were considered nonresponders (NRs).
Results
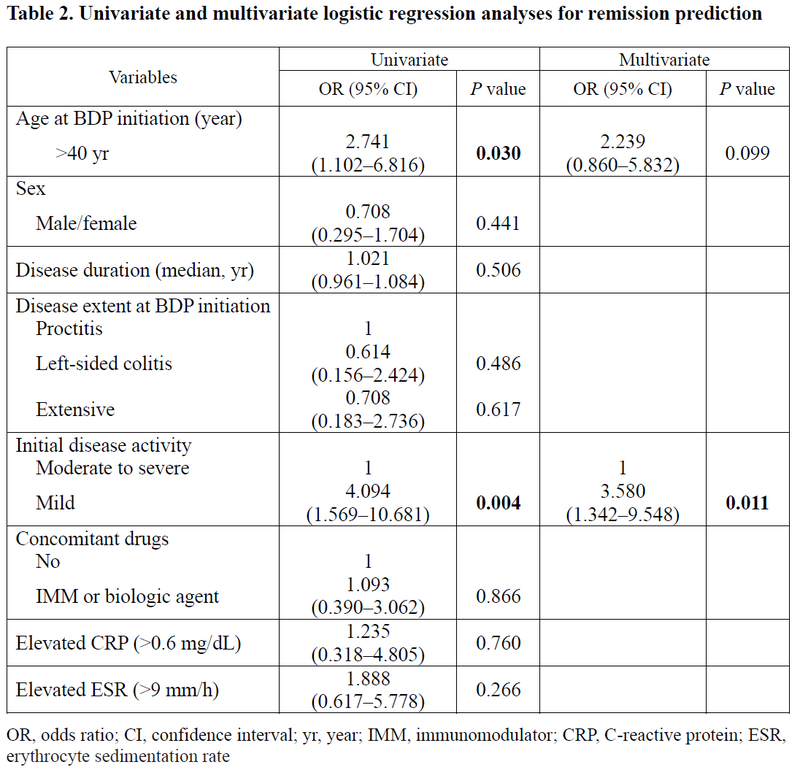
Of total 100 patients, 37 showed CREM, 19 showed CRES, and 44 were NRs (Table 1). The CREM group included more patients with mild disease activity (75.7% vs. 43.2%, P=0.011) than the NR group. In contrast to NR group, responders including CREM and CRES showed significant improvement of posttreatment ESR and CRP (ESR with P=0.001, CRP with P=0.004, respectively). Moreover, initial rectal bleeding subscore (RBS) was significantly different between CREM and CRES, or NR (both with P<0.001). In multivariate logistic regression analysis, initially mild disease activity was the only predictive factor for CREM (odds ratio, 3.580; 95% confidence interval, 1.342–9.548) (Table 2).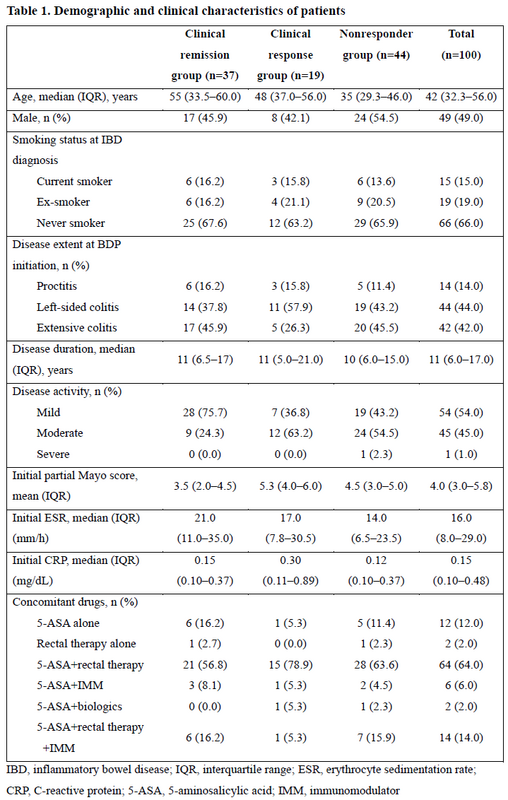
Conclusion
Oral BDP is efficacious as an add-on therapy in Korean UC patients. In case of acute aggravation, patients with mild disease activity, especially low RBS, may be good candidates for oral BDP.


