P480 Ustekinumab Dose Intensification Is Effective in One-Third of Asian Inflammatory Bowel Disease Patients, Results From A Tertiary Inflammatory Bowel Disease Centre.
Lim, C.T.(1)*;Tay, S.W.(1);Lim, G.H.(2);Salazar, E.(1);Ong, W.C.(3);Chan, W.P.W.(1);Tan, M.T.K.(1);
(1)Singapore General Hospital, Department of Gastroenterology and Hepatology, Singapore, Singapore;(2)Singapore General Hospital, Health Services Research Unit, Singapore, Singapore;(3)Singapore General Hospital, Department of Pharmacy, Singapore, Singapore;
Background
Ustekinumab (UST) is an effective biologic for treatment of Inflammatory Bowel Disease (IBD). However, some patients treated with UST have suboptimal clinical response with standard induction and maintenance doses. In some patients, strategies such as dose intensification (DI) and reinduction have been used to improve response. The aims of this study are to identify factors associated with DI for our IBD cohort and also effectiveness of reinduction during DI.
Methods
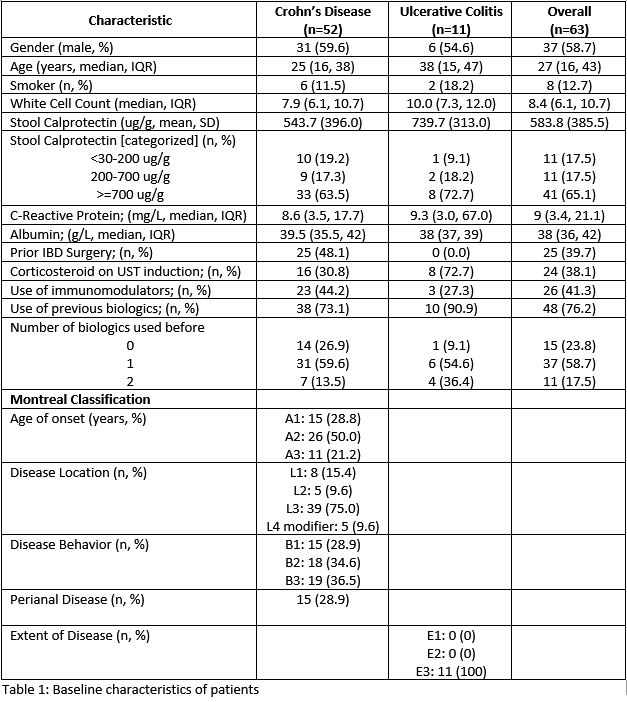
This was a retrospective cohort study on patients with Crohn’s disease (CD) and ulcerative colitis (UC) who received UST from September 2017 to October 2022 in a tertiary center. Patients were identified from a prospectively maintained IBD registry. Patient’s demographics, biochemical profile, disease phenotype, UST intervals and previous use of biologics were retrieved. Statistical analysis was performed to identify factors which are predictive of UST DI and effectiveness of UST DI.
Results
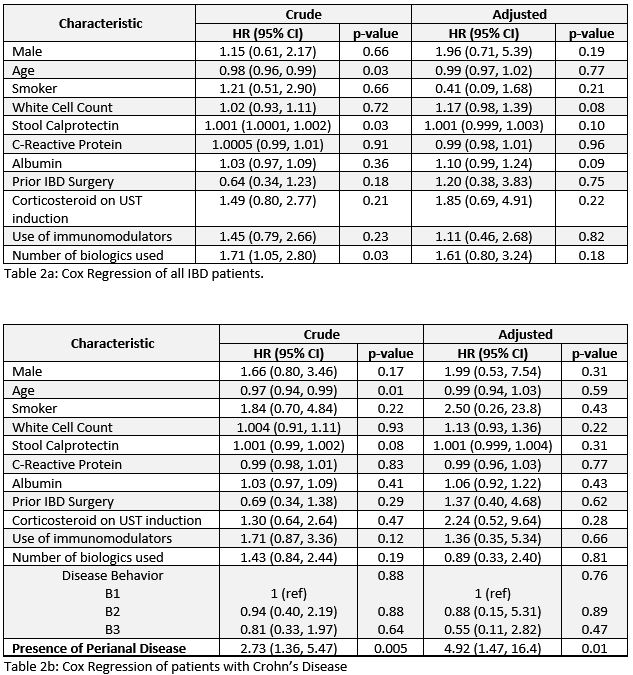
A total of 63 patients with IBD on UST were identified, with most being bio-experienced (76.2%). Forty two patients (34 CD, 8 UC) underwent UST DI to either 6-weekly (n=19, 45.2%) or 4-weekly (n=23, 35.9%). The median duration of UST treatment is 108.0 weeks (IQR 44.7-185.9) while the median time to intensification was 31.1 weeks (IQR 17.8-65.7). Presence of perianal disease in CD (HR 4.9; 1.47 – 16.4) was associated with DI. However, prior usage of biologics (HR 1.61; 0.80 - 3.24) and usage of steroids during induction (HR 1.85; 0.69 – 4.91) were not significant factors for DI.
After DI, there were 14 (33.3%) patients who achieved treatment end points (corticosteroid-free clinical remission, biochemical remission, endoscopic healing (for UC) or transmural healing (for CD) by week 52. In patients who had UST DI, UST re-induction was given to 13 (10 CD, 3 UC) and 4 achieved treatment endpoints but this was not statistically significant (p=0.99). There were 26 patients who failed to achieved end point at week 52 of which 8 patients were continued on UST at dose intensified regime while 11 patients were switched to other biologics.
Conclusion
Two third of IBD patients undergo DI while on UST treatment and the median time to DI is about 6 months after induction. CD patients with perianal disease is more likely to undergo DI. One third of dose intensified patients achieved remission at week 52 while reinduction of UST during DI did not improve outcome for patients.