P486 Improving enhanced recovery after surgery (ERAS): the effect of a patient-centred mobile application on patient participation in colorectal surgery
Van Der Storm, S.(1)*;Jansen, M.(1);Marsman, H.A.(2);Molly, G.P.(3);Consten, E.C.(4);Groot, L.M.(4);Boer, F.C.(5);Corver, M.(5);Oosterling, S.J.(6); Tuynman, J.B.(7);Bemelman, W.A.(1);Buskens, C.J.(1);Schijven, M.P.(1);
(1)Amsterdam University Medical Center- location AMC, Surgery, Amsterdam, The Netherlands;(2)OLVG Hospital, Surgery, Amsterdam, The Netherlands;(3)OLVG, Surgery, Amsterdam, The Netherlands;(4)Meander Medical Center, Surgery, Amersfoort, The Netherlands;(5)Zaans Medical Center, Surgery, Zaandam, The Netherlands;(6)Spaarne Gasthuis Hospital, Surgery, Hoofddorp, The Netherlands;(7)Amsterdam University Medical Center- location VUmc, Surgery, Amsterdam, The Netherlands;
Background
Perioperative care in colorectal surgery is systematically established in the Enhanced Recovery After Surgery (ERAS) protocol. The ERAS protocol improved perioperative colorectal care in a multidisciplinary and multimodal manner, simulating early and safe hospital discharge. In general, compliance with the ERAS protocol is adequate; however, there is still room for improvement in compliance by actively involving patients in their recovery. The main objective of this study was to investigate whether compliance with selected items in the ERAS protocol could be improved by actively involving patients in the ERAS colorectal care pathway using a patient-centred mobile application.
Methods
This multicentre, randomised controlled trial was conducted in accordance with the CONSORT statement. Patients aged 18 years or older who underwent elective colorectal surgery and had a smartphone were included. Patients in the intervention group used a mobile application combined with an activity tracker to be guided and simulated through the ERAS pathway. Patients in the control group received standard care and wore an activity tracker to monitor their daily activities. The primary outcome was overall compliance with the selected active elements of the ERAS protocol. Secondary outcomes included length of hospital stay, number of complications, reintervention, and readmission rates, and patient-reported outcome measures (PROMs) such as quality of life, and patient satisfaction with received care.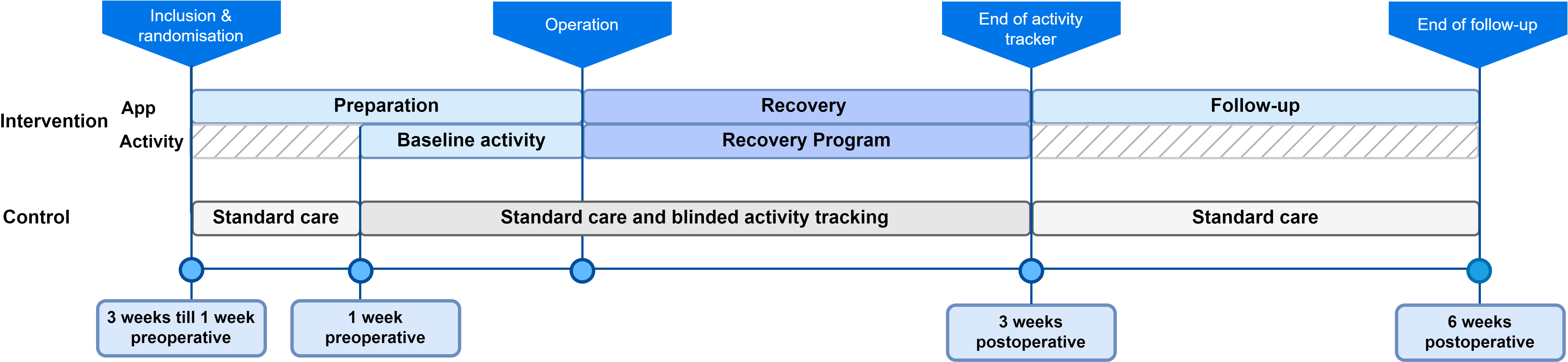

Results
Between October 2019 and September 2022, 170 participants were enrolled and randomised to either the intervention (n=84) or control (n=86) groups. Thirty participants (17.6%) were lost to follow-up and 140 participants were included in the primary outcome analysis. Compliance with the selected active ERAS elements was 76.8% and 63.7% in the intervention and control groups, respectively (p<0.001). Complications or patient reported outcomes did not differ between the groups.
Conclusion
A patient-centred mobile application improved compliance with the active elements of the ERAS protocol. The implementation of this mobile application is recommended for patients undergoing elective colorectal surgery.


