P613 Use of adalimumab biosimilar ABP 501 in Crohn’s disease: A real-life experience
D.G. Ribaldone1, M. Vernero2, R. Pellicano3, M. Morino1, G.M. Saracco4, M. Astegiano3
1Department of Surgical Sciences, University of Turin, Turin, Italy, 2First Department of Internal Medicine, IRCCS Policlinico San Matteo, University of Pavia, Pavia, Italy, 3Unit of Gastroenterology, Molinette Hospital, Turin, Italy, 4Department of Medical Sciences, University of Turin, Turin, Italy
Background
The use of biologics in Crohn’s disease (CD) entails an increasing cost on national health systems. The use of biosimilars of adalimumab in CD is based on the concept of extrapolation of the results obtained in rheumatoid arthritis and in psoriasis, while no study about the efficacy and safety on CD of the biosimilars approved in Europe have been published. The aim of our study was to analyse, for the first time in literature, the effectiveness and safety of ABP 501 in CD patients naïve to adalimumab and its retention rate in CD patients who switched from adalimumab originator.
Methods
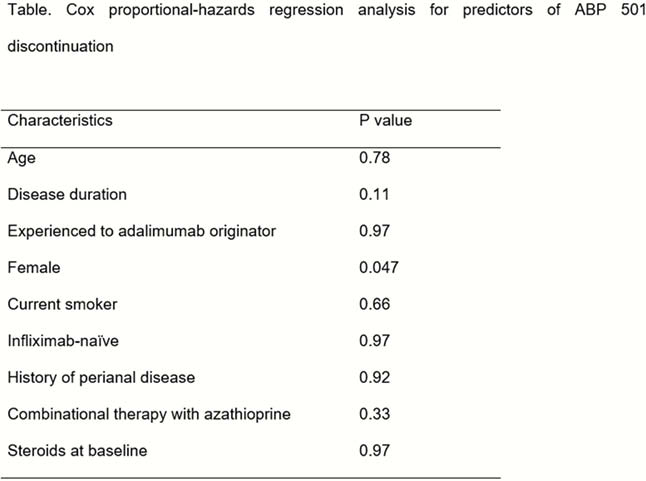
We performed an observational study on patients prospectively followed at the gastroenterology clinic of the Turin University Hospital. Inclusion criteria are (a) CD diagnosed according to ECCO criteria; (b) age ≥16 years; (c) initiation of therapy with ABP 501. Exclusion criterian is follow-up duration of less than 3 months for adalimumab-naïve patients, less than 6 months for patients who switched to ABP 501. Primary outcomes were (a) for patients treated with ABP 501 as first adalimumab: clinical response rate at 12 weeks and (b) for patients who switched to ABP 501: drug retention at 24 weeks. Secondary outcomes were (a) clinical remission rate at week 12 (for patients treated with ABP 501 as first adalimumab); (b) HBI and CRP reduction at week 12 (for patients treated with ABP 501 as first adalimumab), no significant change in HBI and CRP values at week 24 (for patients who switched to ABP 501); (c) analysis of predictors; and (d) adverse events incidence.
Results
Eighty-seven patients were included, of which 25 were naïve to adalimumab originator and 62 were switched to ABP 501. In adalimumab-naïve patients, the clinical response at 3 months was 60% (15/25), clinical remission at 3 months was 56% (14/25). At 6 months, 95.2% (59/62) of the patients switched to ABP 501 were still in therapy, without a significant increment of clinical activity (Harvey–Bradshaw Index from 3.4, 95% CI = 2.4 – 4.4, to 3.8, 95% CI = 2.7 – 4.9, p = 0.23), and inflammatory biomarker (CRP from 4.2 mg/l, 95% CI = 2.5 mg/l – 5.9 mg/l, to 3.6 mg/l, 95% CI = 2.2 mg/l – 5 mg/l, p = 0.32). No unexpected adverse events occurred during the study period.
Conclusion
Our results support ABP 501 as an efficacious and well-tolerated drug, at least in the short-term, and its interchangeability with its originator in the treatment of CD.


