P644 Hospitalization and abdominal surgery rates in CD according to drug-dispensing: a temporal trend analysis from the Brazilian public healthcare national system
Magro, D.O.(1);Kotze, P.G.(2);Quaresma, A.B.(3);Damiao, A.O.M.C.(4);Valverde, D.A.(5);Panaccione, R.(6);Coward, S.(7);Ng, S.C.(8);Kaplan, G.G.(7);Coy, C.S.R.(1);
(1)University of Campinas UNICAMP, Colorectal Surgery Unit, Campinas, Brazil;(2)Catholic University or Paraná PUCPR, IBD outpatient Clinics- Colorectal Surgery Unit, Curitiba, Brazil;(3)UNOESC, Colorectal Surgery Unit, Joaçaba, Brazil;(4)University of Sao Paulo, Department of Gastroenterology, Sao Paulo, Brazil;(5)Techtrials Healthcare, Data Science, Vinhedo, Brazil;(6)University of Calgary, Division of Gastroenterology and Hepatology, Calgary, Canada;(7)University of Calgary, Department of Medicine and Community Health Sciences, Calgary, Canada;(8)Chinese University of Hong Kong, Department of Gastroenterology, Hong Kong, Hong Kong- China
Background
The impact of current medical options in Crohn’s disease (CD) on hospitalization and surgical rates may be conflicting, and there is lack of data in newly industrialized countries. This study aims to describe temporal trends of proportional hospitalization and CD-related abdominal surgery rates according to drug-dispensing in Brazil, using public healthcare datasets.
Methods
All CD patients from the unique public healthcare national system (DATASUS) were included from January 2012 to December 2020 and identified according to ICD codes, medication or CD-related procedures. Data extraction was performed with the platform “TT Disease Explorer” (Techtrials Healthcare Data Science), which collects publicly available data via electronic algorithms with automatic updates. Annual rates of all-cause hospitalization and CD-related abdominal surgical procedures were captured and stratified by type of drug dispended. Average Annual Percent Change (AAPC) and 95% confidence intervals (CI) were calculated using poisson (or negative binomial) regression.
Results
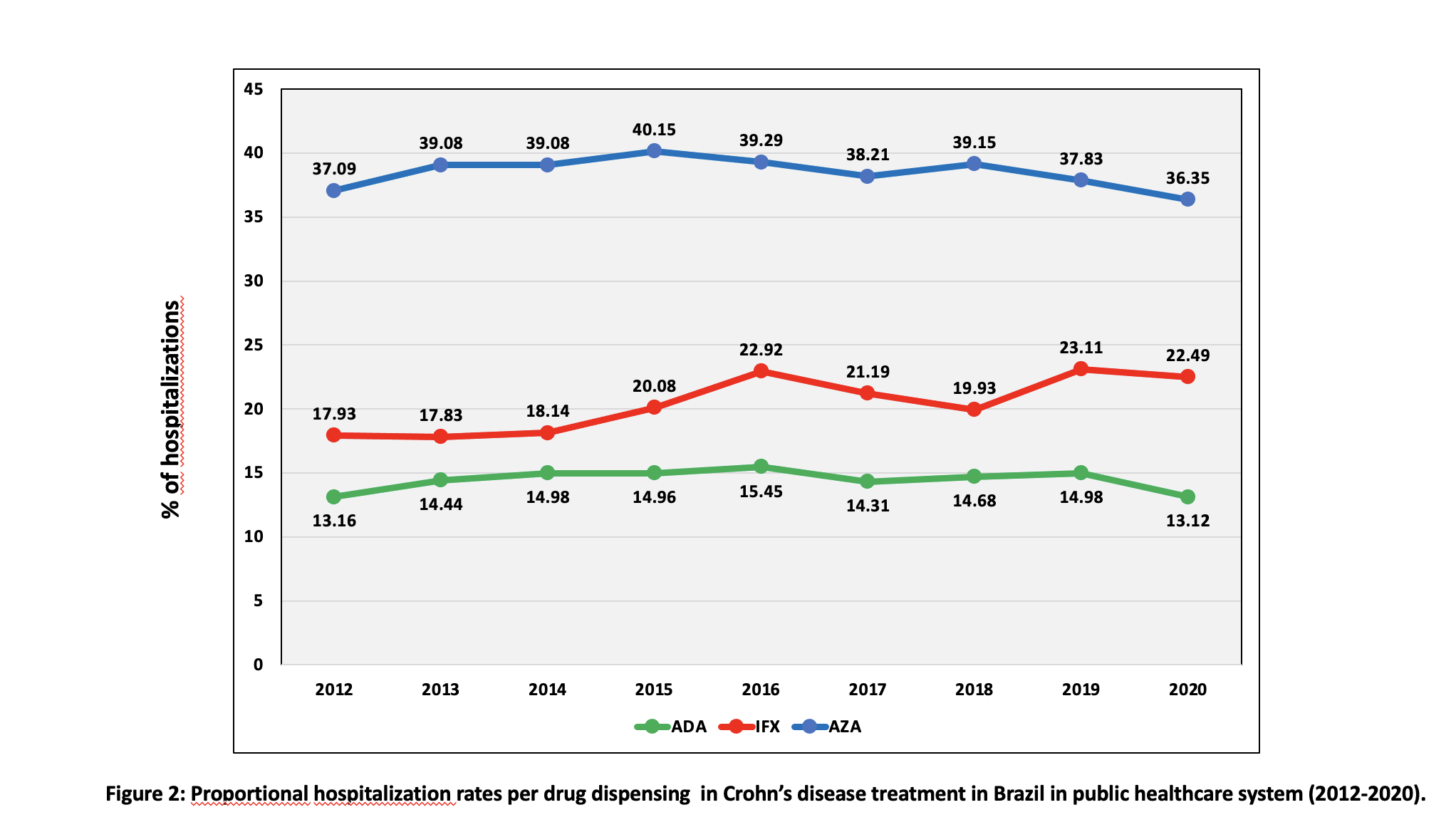
The absolute number of registries of overall drug-dispensing for CD was 178,209, being 32.03% for Azathioprine (AZA), 10.91% for infliximab (IFX) and 10.52% for Adalimumab (ADA). AZA dispensing increased from 28.60% to 30.83% (AAPC 1.15; CI 0.23-2.09; p=0.015), ADA increased from 5.98% to 12.03% (AAPC 8.79; CI 6.33-11.30; p<0.001) and IFX increased from 7.09% to 12.03% (AAPC 7.52; CI 6.94-8.10; p<0.001) (figure 1). A total of 39,161 hospitalizations (all-cause) were captured in the same period. Hospitalization rates with AZA varied from 37.09% to 36.35% (AAPC -0.42, CI -1.08-0.24; p=0.209); for ADA remained stable (13.16% to 13.12%, AAPC -0.03; CI -1.10-1.05; p=0.962) and for IFX increased from 17.93% to 22.49% (AAPC 3.21; CI 1.66-4.79, p<0.001) (figure 2). Regarding CD-related abdominal surgical procedures (n=1181), rates were stable for AZA (AAPC 1.34; CI -8.41-12.12; p=0.797). Considering the use of anti-TNF agents, rates were stable with ADA, varying from 26,7% to 20,0% (AAPC -1.64; CI -13.84-12.29; p=0.807) and decreased from 33,3% to 4,5% for IFX (AAPC -17.05; CI -28.19- -4.17; p=0.011) (figure 3).


Conclusion
In this large national study, there was an increase in the number of dispensings of AZA, IFX and ADA for CD from 2012-2020 in the public healthcare system in Brazil, due in part to the increasing prevalence of CD. All-cause hospitalization rates remained stable for AZA and ADA, and increased in IFX patients. A reduction in CD-related abdominal surgical procedures was observed in patients who used IFX and were stable with AZA and ADA. These data can be used for future strategic planning in the national public healthcare system (SUS) in CD management in Brazil.


