P778 Effectiveness and safety of a second-line rescue therapy for acute severe ulcerative colitis refractory to infliximab or ciclosporin (REASUC study)
García, M.J.(1)*;Riestra, S.(2);Amiot, A.(3);Julsgaard, M.(4);García de la Filia, I.(5);Calafat, M.(6);Aguas, M.(7);de la Peña, L.(8);Roig-Ramos, C.(9);Caballol, B.(10);Casanova, M.J.(11);Farkas, K.(12);Boysen, T.(13);Bujanda, L.(14);Cuarán, C.(15);Dobru, D.(16);Fousekis, F.(17);Gargallo-Puyuelo, C.J.(18);Savarino, E.(19);Calvet, X.(20);Huguet, J.M.(21);Kupcinskas, L.(22);López-Cardona, J.(23);Raine, T.(24);van Oostrom, J.(25);P. Gisbert*, J.(11);Chaparro*, M.(11);
(1)Hospital Universitario Marqués de Valdecilla- IDIVAL- Universidad de Cantabria, Gastroenterology and Hepatology, Santander- Cantabria, Spain;(2)Hospital Universitario Central de Asturias- Instituto de Investigación Sanitaria del Principado de Asturias ISPA, Gastroenterology, Oviedo, Spain;(3)CHU Bicêtre- Universite Paris Saclay, Gastroenterology, Paris, France;(4)Aarhus University Hospital, Gastroenterology and Hepatology, Aarhus, Denmark;(5)Hospital Universitario Ramón y Cajal, Gastroenterology, Madrid, Spain;(6)Hospital Universitari Germans Trias i Pujol- Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas CIBEREHD, Gastroenterology, Barcelona, Spain;(7)La Fe University and Polytechnic Hospital- Health Research Institute La Fe, Gastroenterology, Valencia, Spain;(8)Bellvitge University Hospital, Gastroenterology, Barcelona, Spain;(9)Hospital de la Santa Creu i Sant Pau, Digestive Services, Barcelona, Spain;(10)Hospital Clinic- Institut d'Investigacions Biomèdiques August Pi i Sunyer IDIBAPS- Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas CIBEREHD, Gastroenterology, Barcelona, Spain;(11)Hospital Universitario de La Princesa- Instituto de Investigación Sanitaria Princesa IIS-Princesa- Universidad Autónoma de Madrid UAM and Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas CIBEREHD, Gastroenterology, Madrid, Spain;(12)Universitary of Szeged, Medicine, Szeged, Hungary;(13)Medical Section- Copenhagen University Hospital Hvidovre, Gastrounit, Hvidovre, Denmark;(14)Biodonostia Health Research Institute- Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas CIBEREHD- Universidad del País Vasco UPV/EHU, Gastroenterology, San Sebastián, Spain;(15)Hospital Universitario Miguel Servet, Gastroenterology, Zaragoza, Spain;(16)University of Medicine and Pharmacy- Science and Tehnology "G E Palade" Tg.Mures, Gastroenterology, Târgu-Mureș, Romania;(17)University Hospital of Ioannina, Gastroenterology, Ioannina, Greece;(18)University Clinic Hospital Lozano Blesa- Institute for Health Research Aragón IIS Aragón, Gastroenterology, Zaragoza, Spain;(19)Azienda Ospedale Università di Padova AOUP- Department of Surgery- Oncology and Gastroenterology DiSCOG- University of Padua, Gastroenterology, Padua, Italy;(20)Servei d'Aparell Digestiu. Corporació Sanitària Universitària Parc Taulí- Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas CIBEREHD- Instituto de Salud Carlos III- Universitat Autònoma de Barcelona, Gastroenterology, Sabadell, Spain;(21)General University Hospital of Valencia, Digestive Diseases, Valencia, Spain;(22)Institute for Digestive Research- Lithuanian University of Health Sciences, Gastroenterology, Kaunas, Lithuania;(23)Guy's and St Thomas' Hospital, IBD Unit, London, United Kingdom;(24)Cambridge University Hospitals NHS Foundation Trust, Gastroenterology, Cambridge, United Kingdom;(25)Universitair Medische Centra UMC, Gastroenterology and Hepatology, Amsterdam, The Netherlands;
Background
The advent of new therapeutic agents and the improvement of supporting care might change the management of acute severe ulcerative colitis (ASUC) to avoid colectomy and change the natural history of the disease. The effectiveness and safety of sequential treatment should be evaluated in this scenario.
Main aim: To evaluate colectomy-free survival in patients with ASUC refractory to intravenous steroids who had failed infliximab (IFX) or ciclosporin (CyA) and received a second salvage therapy. Secondary aims: To evaluate the short-term and long-term effectiveness and to assess the safety of this strategy.
Methods
Multicentre study of patients admitted with ASUC refractory to corticosteroids who failed CyA or IFX and had received a second salvage therapy during the same hospital stay. Clinical activity was classified based on partial Mayo score and Lichtiger activity index. Patients who stopped second-line therapy due to disease activity or adverse events were considered as failures. Short-term colectomy-free survival was assessed by logistic regression analysis while long-term colectomy-free survival was evaluated by Kaplan-Meier curves and Cox regression models.
Results
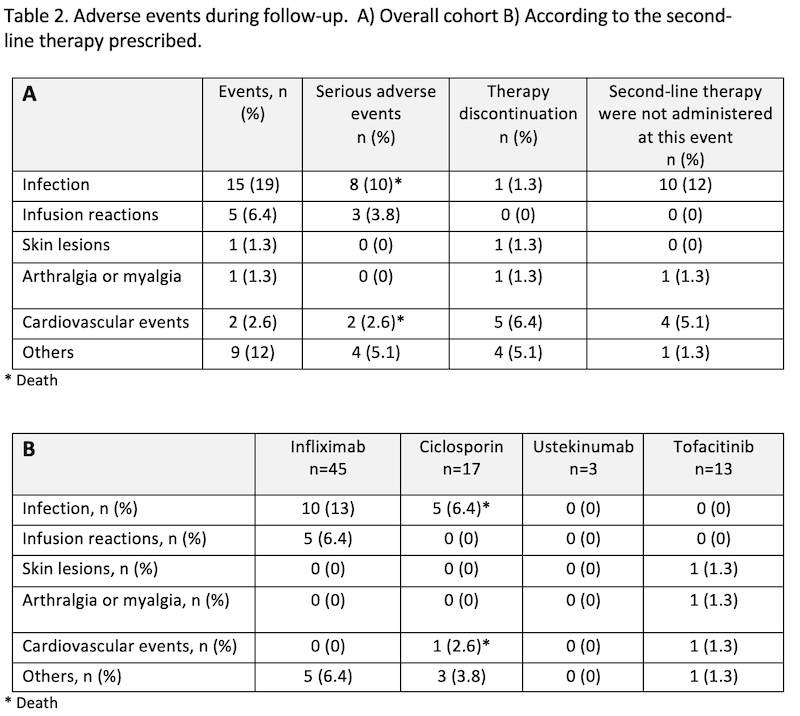
Results: A total of 78 patients from 24 centres were included. As first-line therapy, IFX was administered to 32 patients, and CyA to 46. Baseline characteristics are detailed in table 1. The prescribed salvage therapy was IFX in 45 patients, CyA in 17, tofacitinib in 13, and ustekinumab in 3 (Figure 1). Colectomy was required in 29 patients (37%) during the follow-up (median 14 days, IQR 3-23). Colectomy-free survival curves are shown in figure 2-A and 2-B. Second-line salvage therapy with CyA was associated with a higher rate of colectomy (Figure 2-C). A total of 39 patients (55%) at week 12 and 20 patients (33%) at month 12 had clinical response; 31 patients (44%) at week 12 and 18 (30%) at month 12 were in clinical remission. At the last visit of the study, 26 patients (33%) were still under the second-line therapy. Adverse events (AE) were recorded in 26 patients (33%), being serious in 15. Resolution with no sequelae was observed in 20 patients (77%). No differences in AE between the second-line therapies prescribed were observed. There were two deaths: one due to cerebral thrombophlebitis and another due to bacteriemia after colectomy, both when CyA was the salvage therapy prescribed (Table 2). 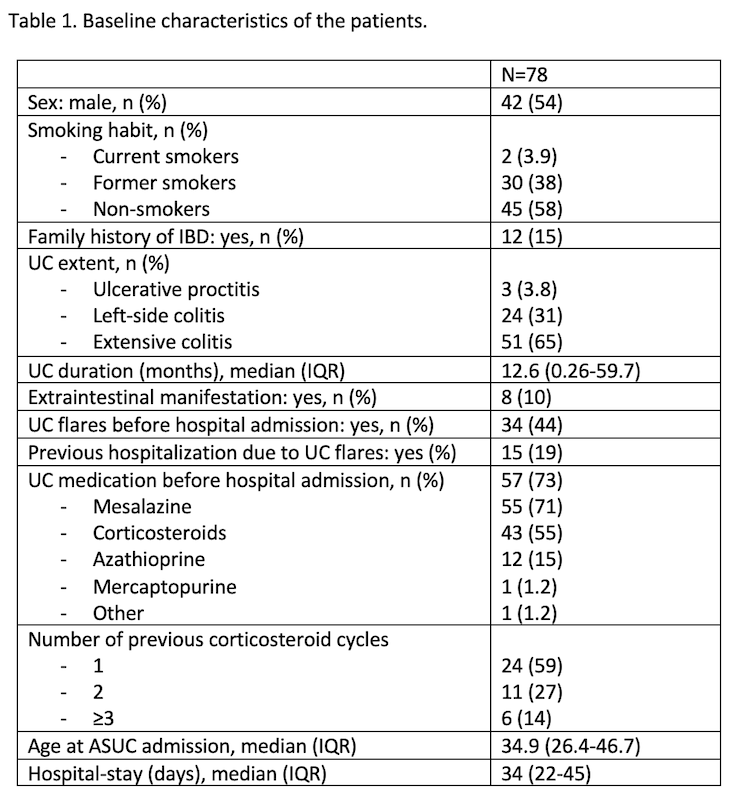


Conclusion
A second-line salvage therapy avoids colectomy in a considerable percentage of patients admitted by ASUC and therefore it could be considered after IFX or CyA failure. The individual therapeutic approach must be discussed with patients due to the risk of serious adverse events.


