ECCO Fellowship Study Synopsis: Andres Machicote
Andres Machicote, ECCO Fellowship Awardee
Targeting CD4+ T-cell plasticity in Inflammatory Bowel Diseases
 Andres Machicote Andres Machicote© Andres Machicote |
Aim of research
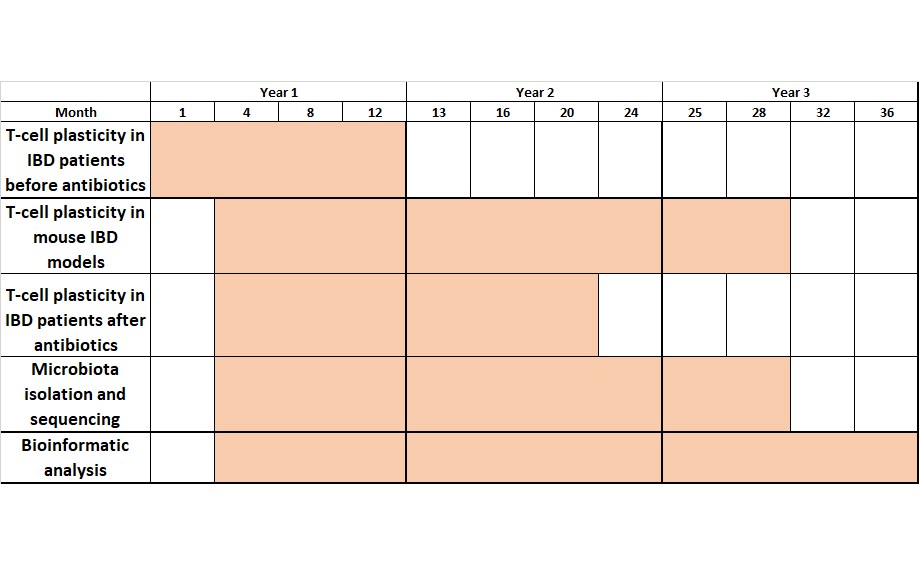
Inflammatory Bowel Diseases (IBD) are devastating diseases induced by a breakdown of gut homeostasis, in which CD4+ effector T cells are major mediators of the inflammatory response. Interestingly, CD4+ effector T cells can convert into regulatory T cells, thereby controlling inflammation. However, the forces driving this T-cell plasticity remain largely unexplored, especially in IBD patients. In this regard, changes in the microbiota that can induce different T-cell profiles are commonly observed during IBD. The present proposal aims to elucidate how CD4+ T-cell plasticity can be modulated from a pro-inflammatory towards an anti-inflammatory profile in IBD patients. Our hypothesis is that the microbiota may modulate T cells towards an anti- or pro-inflammatory profile. To address this issue, we aim to analyse T-cell plasticity in IBD patients and mouse IBD models, and to correlate our findings with the microbiota composition. Furthermore, we will study whether antibiotics commonly used to treat IBD affect T-cell plasticity through microbiota-dependent processes.
Methods
To achieve our goals, we will first study CD4+ T-cell plasticity in IBD patients and mouse IBD models, such as reporter and IBD-microbiota reconstituted germ-free mice. Additionally, we will use the T-cell receptor as a barcode, to follow up and analyse at a single cell level the T-cell immune profiles before and after antibiotic treatment. Furthermore, to determine whether T-cell plasticity is modulated by the microbiota, we will sequence the intestinal flora.
Anticipated main impact
Current IBD therapies are palliative in character and do not provide a cure. Thus, new and more specific therapies that aim to restore immune homeostasis are badly needed. The conversion of effector T cells into regulatory T cells is a tempting approach to counteract intestinal inflammation in IBD patients. Therefore, identifying the forces driving this process towards an anti-inflammatory profile could boost the development of safer strategies, hence diminishing the side effects associated with the current immunosuppressive treatments.
Proposed timeline